Introduction
The coronavirus disease 2019 (COVID-19) caused more than 6 million deaths worldwide (1), and vaccination remains to be the most effective way to reduce the deaths in addition to infection control measures in the community. The Turkish Ministry of Health offered two types of vaccines by January 13, 2021, CoronaVac (Sinovac Biotech, China), and three months later, by April 12, 2021, Pfizer-BioNTech (Pfizer-BioNTech). Because of having two different types of vaccines, there was an opportunity to choose between Pfizer-BioNTech or CoronaVac vaccines. Meanwhile, for those who had already received two doses of CoronaVac, another booster dose of CoronaVac or two doses of Pfizer-BioNTech were offered. The decision to select one of two options sometimes caused confusion among the people and healthcare workers. Having two vaccines in Turkey allowed us to compare the outcomes of using different vaccines.
A recent meta-analysis, including the community-level studies, reported that the Pfizer-BioNTech vaccine had 91.2% effectiveness while CoronaVac had 65.7% (2). Our study aimed to describe the impact of the CoronaVac and Pfizer-BioNTech vaccines on outcomes among hospitalized patients during a six-month period.
Materials and Methods
Study Design and Study Population
RT-PCR confirmed >18 years of age patients who were hospitalized between June 2021 and December 2021 were included. Having the 15% fatality rate among unvaccinated inpatients from previous data and considering a 10% reduction of fatality rate to 5%, with type I error of 5% and type II error of 20%, we calculated the sample size as 282 patients.
Age, gender, body mass index (BMI), admission and discharge dates, comorbidities, tocilizumab, anakinra, and dexamethasone use were retrieved from the medical records. The patients who had missing data were excluded. In addition, two patients having Janssen COVID-19 vaccines (Johnson&Johnson) were excluded from the data for the sake of comparison. The patients were categorized as mild, moderate, severe, and critical according to the World Health Organization (WHO) classification (3). Full vaccination was defined as being vaccinated at least two weeks passed after the second dose of vaccines.
Koç University Committee on Human Research and Institutional Review Board approved the ethical considerations about study (reference number 2020.145.IRB1.034). Informed consent was obtained from the patients.
Statistical Analysis
Comparison of categorical variables was performed by chi-square test and continuous variables by t-test. Univariate and multivariate analysis for the fatality predictors was performed using logistic regression. The statistical significance was set as p<0.05.
Results
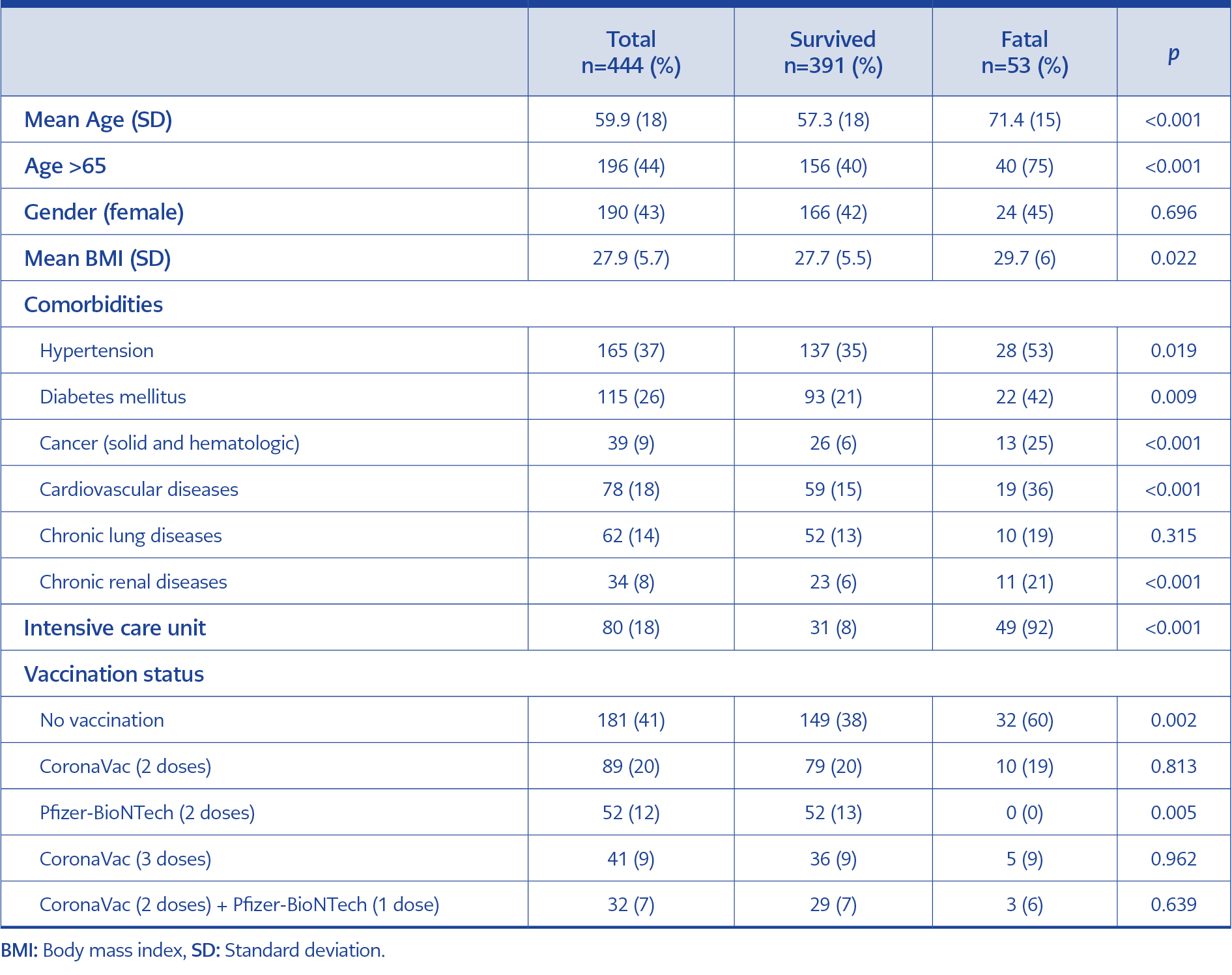
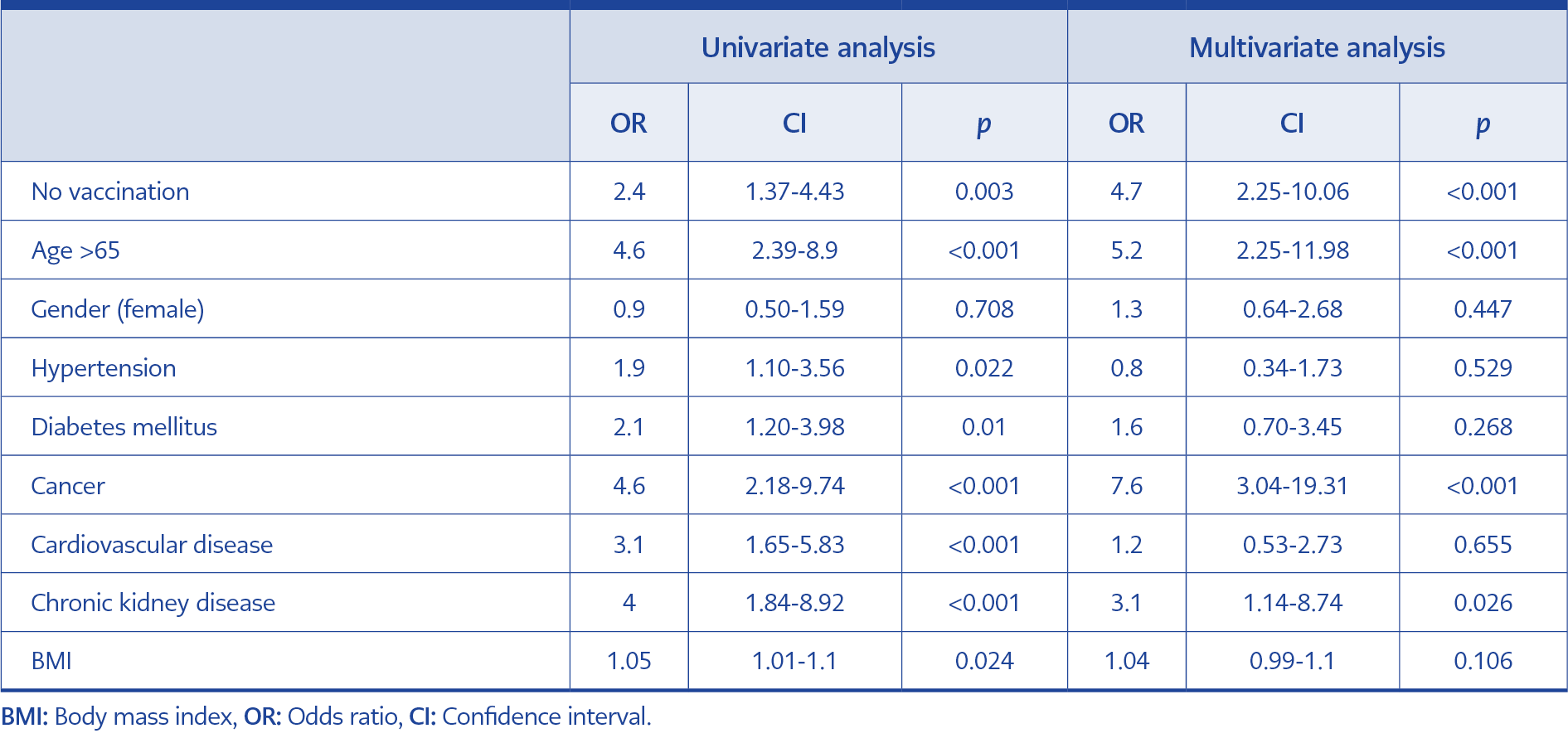
In total, 444 RT-PCR confirmed >18 years of age inpatients were included (46.6% at American Hospital, 53.4% at Koç University Hospital). The mean age of the patients was 59.9, 44% were older than 65 years of age, and 42.8% were female (Table 1). The most common comorbidity was hypertension (39%), followed by diabetes mellitus (27%), cardiovascular diseases (18.4%), chronic lung diseases (14.6%), cancer (9.2%), and chronic renal diseases (8%). Univariate analysis showed that no vaccination (OR=2.4, CI=1.37-4.43; p=0.003), age >65 (OR=4.6, CI=2.39-8.90; p<0.001), hypertension (OR=1.9, CI=1.10-3.56; p=0.022), diabetes mellitus (OR=2.1, CI=1.20-3.98; p=0.010), cancer (OR=4.6, CI=2.18-9.74; p<0.001), cardiovascular diseases (OR=3.1, CI=1.65-5.83; p<0.001), and chronic kidney disease (OR=4.1, CI=1.84-8.92; p<0.001) significantly increase mortality in COVID 19 patients. The fatality rate among the patients who stayed in ICU was 92.4%, and it was 95% among the patients intubated at Koç University Hospital (p<0.001) (Table 2).
In total, 181 patients (40.8%) were unvaccinated before admission, constituting 60% of the fatal cases (p=0.002). None of the patients who were vaccinated with two doses of Pfizer-BioNTech vaccines died (p=0.005). However, the booster dose of Pfizer-BioNTech after two doses of CoronaVac has no clinical significance for preventing mortality (p=0.639).
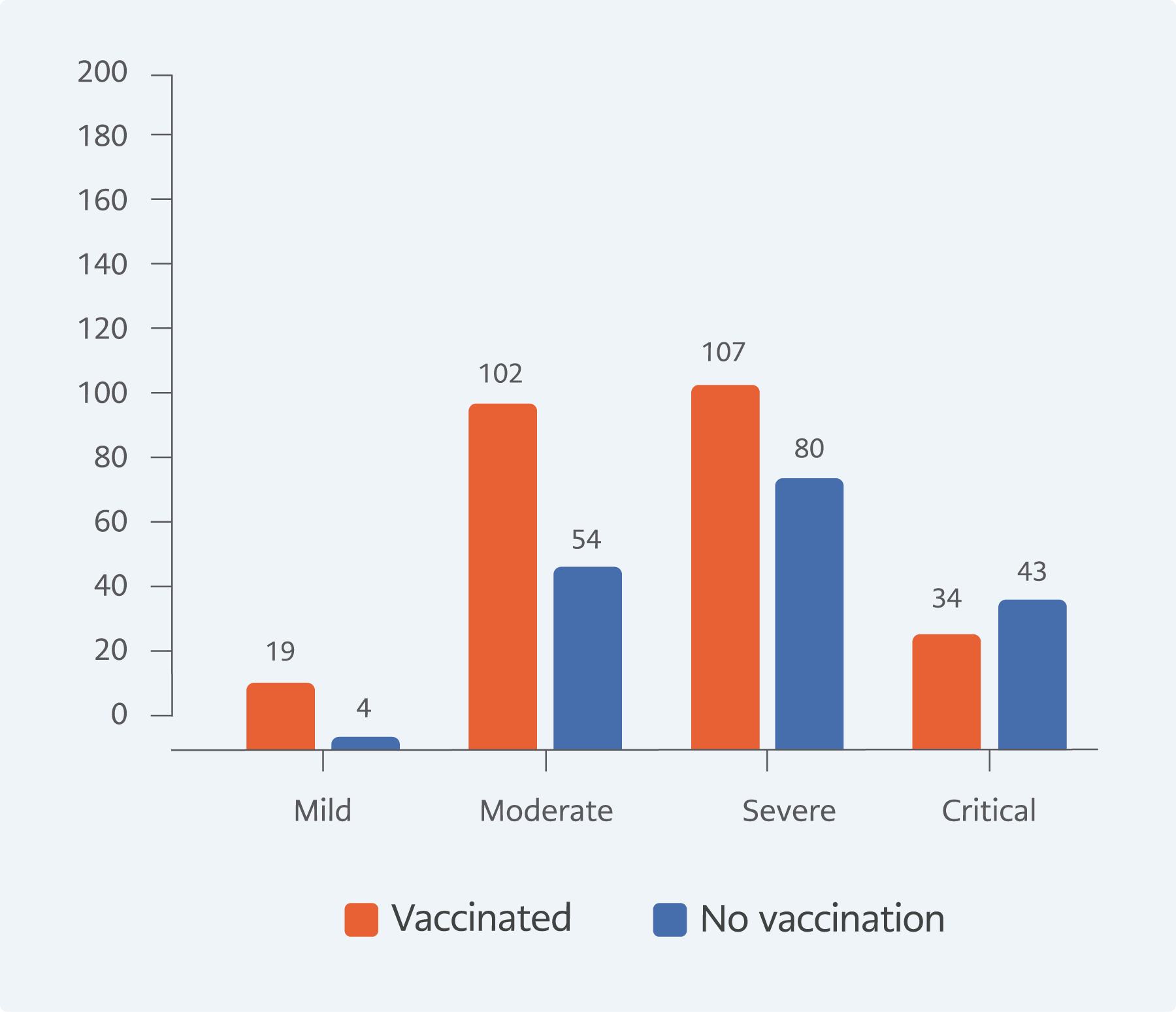
The proportion of critically ill patients in the unvaccinated group was 23.8%, whereas it was 12.9% among the patients who received at least one type of vaccine (p=0.002, Figure 1).
In multivariate analysis for the fatality prediction, unvaccinated patients were 4.7 times more likely to die than vaccinated patients (OR=4.7, CI=2.25-10.06; p<0.001). Besides being unvaccinated, other significant predictors of fatality were age >65, cancer, and chronic kidney disease (Table 2).
Discussion
The clinical observations reflecting real-life experiences are important to provide more convincing information about the COVID-19 vaccines. In this study, we described the clinical impact of having Pfizer-BioNTech or CoronaVac vaccines. We approved the findings of in vitro studies which showed that the Pfizer-BioNTech vaccine had higher effectiveness than CoronaVac. In previous in vitro studies, the neutralizing antibody levels after three months of the Pfizer-BioNTech booster were higher than the antibody levels after CoronaVac in fully, at least two doses of the vaccine according to WHO recommendations, vaccinated individuals (4, 5, 6). In our study, two doses of the Pfizer-BioNTech vaccine were 100% protective against mortality among hospitalized patients because of COVID-19. Since the Pfizer-BioNTech vaccine was 100% protective and predicted the outcome, we did not include it in the multivariate model for mortality.
After the introduction of the Pfizer-BioNTech vaccine on April 12, 2021, there were two options for booster doses after two doses of the CoronaVac vaccine. Studies performed in diverse populations suggested that both Pfizer-BioNTech and CoronaVac booster doses were effective against mortality, and heterologous boosters were found to be superior to homologous boosters in terms of IgG production (7, 8). In our study, a booster dose of Pfizer-BioNTech after two doses of CoronaVac was not found to be preventive against mortality (p=0.639); however, this result might be related to the small sample size in our study.
A study among hospitalized patients with COVID-19 from Turkey reported that full vaccination with CoronaVac or Pfizer-BioNTech was protective against mortality (9), and another study among 1401 hospitalized patients reported Pfizer-BioNTech was more effective than CoronoVac in terms of protection from mortality (10). Vaccines are not only protective against mortality but also decrease disease severity as well as hospital admission. Unvaccinated patients have higher ICU admission, which means critical disease and mechanical ventilation use, compared with vaccinated inpatients in our study.
A Turkish study shows that 53% of unvaccinated patients were admitted to the ICU due to disease severity, which decreased while vaccine doses increased (10). A case-control study of 1197 inpatients showed that the Pfizer-BioNTech vaccine decreased disease severity significantly, and the vaccinated group had lower ICU admission than unvaccinated patients, 24.6% and 40.1%, respectively (p<0.001) (11). Regardless of the type of the vaccine, mechanical ventilation needs, or death was shown significantly lower (p<0.001) among vaccinated patients in a multicenter study. It was 24.9% among vaccinated groups and 40.5% among unvaccinated controls (12). In our study, we do not have the data of non-hospitalized patients, but a comprehensive study from England with more than 150,000 patients included showed that one dose of the Pfizer-BioNTech vaccine had protection against symptomatic disease and reduced the risk of emergency hospital admission by 43% (33% to 52%) (13). In addition, another large study from the UK showed that vaccinated patients were hospitalized less and experienced symptoms of the disease less than others (14). More than 80% percent of the hospitalized patients in our study were either unvaccinated or vaccinated with two doses of CoronaVac.
In our multivariate analysis, being older than 65 years of age, having cancer, and having chronic kidney disease were found to be significant predictors of COVID-19-related fatality. Above-normal inflammatory responses causing higher mortality in older adults may stem from impaired immune system defects increasing with age (9, 15-17). The higher mortality rates in patients with comorbidities may stem from lower vaccine efficacy and risk of morbidity exacerbation (9).
Cancer was a significant contributor in both univariate and multivariate analyses. Patients with cancer are considered highly vulnerable and prone to developing severe COVID-19 symptoms, possibly due to an immunosuppressive state caused directly by the growing tumor itself or indirectly by antineoplastic agents (18). Besides, antineoplastic agents that our patients were using could cause impaired vaccine response, which explains higher mortality. Chronic kidney disease is found to be an important contributor to COVID-19 mortality, the mechanism of which, however, is yet to be found (19). However, it was reported that the patients requiring hemodialysis have lower antibody levels than controls (19). Therefore, it may explain lower vaccine response among patients with chronic kidney disease.
In our univariate analysis, hypertension and diabetes were found to be significantly associated with higher mortality rates; however, in multivariate analysis, these were not significantly associated with mortality. Recent findings showed that hypertension is one of the most common comorbidities in patients with COVID-19, whose prevalence ranges between 10% to 34% (20). However, there is still no consensus about the impact of hypertension on COVID-related mortality (20). Similar to hypertension, the effect of diabetes mellitus on mortality still lacks evidence (21). Although its effect on mortality was reported (22), we did not detect a significant impact in the multivariate analysis.
The major limitation of this study is its retrospective observational design. Our sample size was limited to the number of patients hospitalized in our two hospitals, but we reached statistical significance. We did not include the variants of the SARS-CoV data; however, during the study period, the circulating variants were Delta and Omicron. It is reported that patients who received inactive vaccines are protected from severe disease, and an additional third booster dose is effective (23, 24). In contrast, third doses of CoronaVac were found to be ineffective against severe and fatal diseases in our study. However, the low number of patients with three doses may have an impact on our results.
Among the inpatients with COVID-19, the predictors for mortality were being unvaccinated, older age, cancer, chronic kidney disease, and cardiovascular diseases. Among the vaccinated inpatients, having two doses of the Pfizer-BioNTech vaccine was the only effective protective measure against mortality; however, two doses of the CoronaVac vaccine had no significant effect in preventing fatality.