Introduction
Patients with human immunodeficiency virus (HIV) infection or AIDS are more likely to have diseases that would not usually cause illness in a person with a healthy immune system, called opportunistic infections. Although significant efforts to improve progression along the HIV care continuum, there is still a considerable number of patients who initially present with advanced disease (1). In terms of opportunistic infections, significant differences exist in different parts of regions; for instance, tuberculosis case notification rates were 5-10 times higher in Eastern compared with Western Europe (2). Among 2953 patients with a new HIV diagnosis in our country between 2011 and 2016, 29% of the cases presented with a CD4+ T lymphocyte count of >500 cells/mm3, whereas 46.7% presented with a count of <350 cells/mm3 (3). Similarly, among 693 patients diagnosed in 2011 and 2012, the proportion of patients presenting with advanced HIV disease was 30.6%, while 52.4% were late presenters (4).
This systematic review aimed to investigate opportunistic infections among HIV-infected patients in Turkey.
Methodology and Search Strategy
We searched the Pubmed, Web of Science (WoS), and ULAKBİM TR Dizin databases until October 01, 2022, for the medical subject headings (MeSH) terms of (HIV) OR (AIDS) OR (human immunodeficiency virus) AND (opportunistic infections) AND (Turkey). In addition, we reviewed the references of included articles.
Study Selection
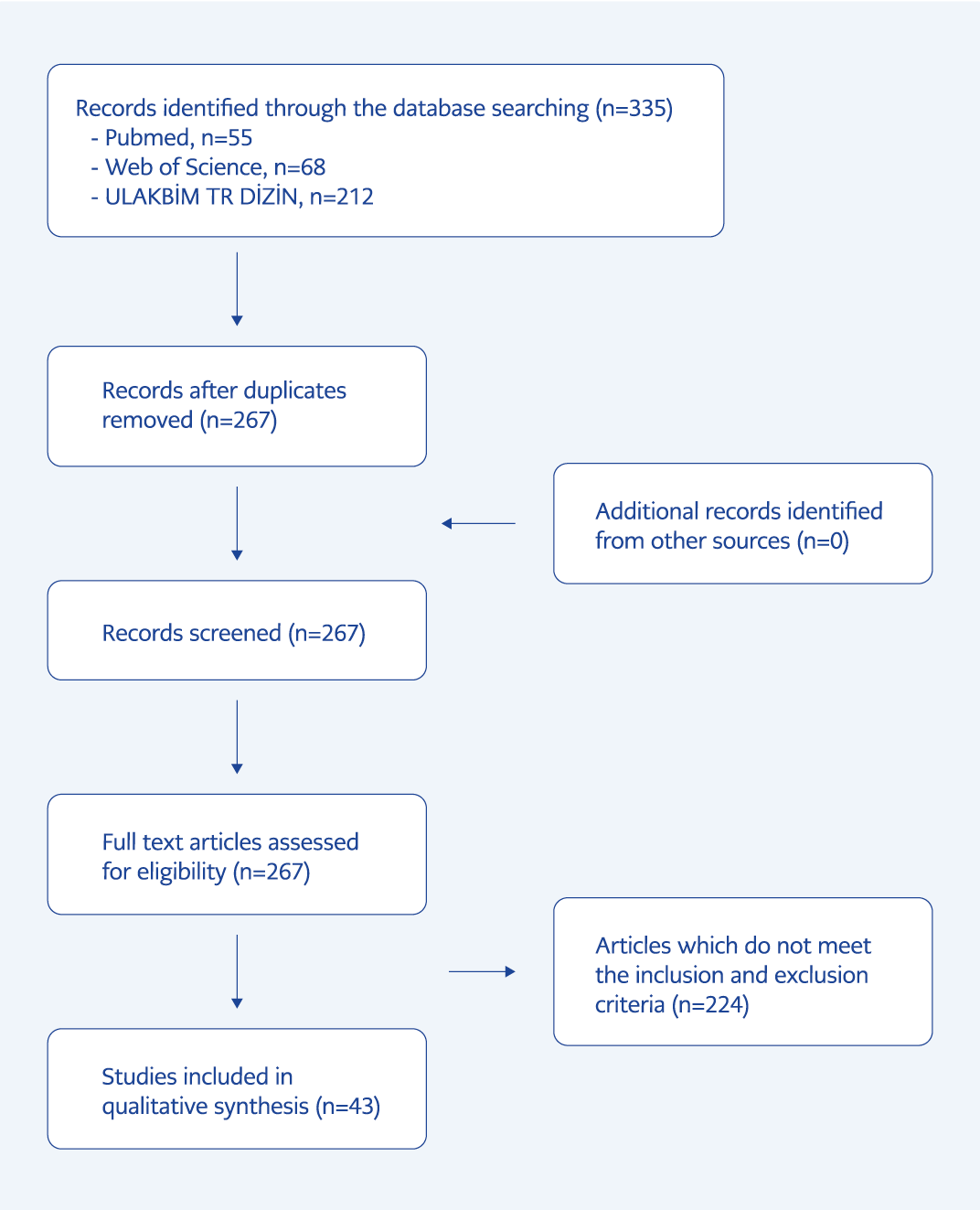
We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement in our study (5, 6). PRISMA flow diagram is presented in Figure 1. We extracted data onto a study-specific form, including the list of clinical questions under the PICO (Population-Intervention-Comparison-Outcome) framework format (Table 1). Studies were initially selected according to the title and abstract screening, and the full text of each potentially eligible study was obtained. Then we selected studies according to inclusion and exclusion criteria.
We included adult (>18-year-old) patients and thestudies met all of the PICO components (Table 1). We excluded the studies which have only seroprevalence data rather than clinical features and laboratory results of the HIV/AIDS patients, the studies about HIV/AIDS patients with comorbidities or co-infections such as syphilis, hepatitis or malignancies, and the studies about opportunistic infections such as pneumonia, diarrhea, wasting syndrome due to HIV, invasive cervical cancer, HIV-related encephalopathy, Kaposi sarcoma or lymphoma that were listed in Centers for Disease Control and Prevention (CDC) common opportunistic infections (7).
Synthesis of Results
All outcomes were analyzed, including the authors, study design, total number of patients, and the assessment of clinical features and laboratory results of the HIV/AIDS patients with an opportunistic infection. The results are also sorted by number of patients respectively. The reference lists of all relevant studies were also checked. Late presentation was defined as a CD4+ T lymphocyte count below 350 cells/μL or presenting with an AIDS-defining event (8). The study was designed as a systematic review, so no ethical committee permission was needed.
Clinical and Research Consequences
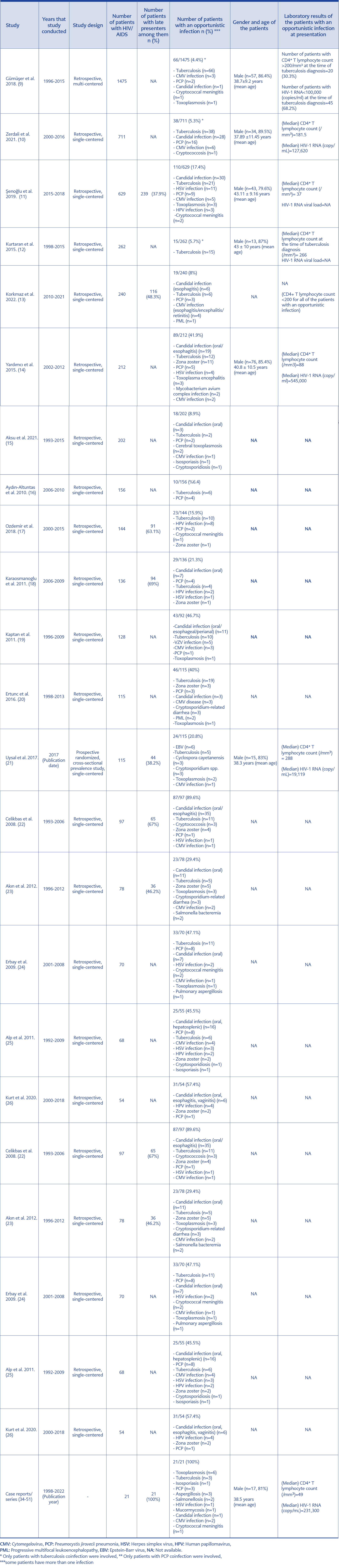
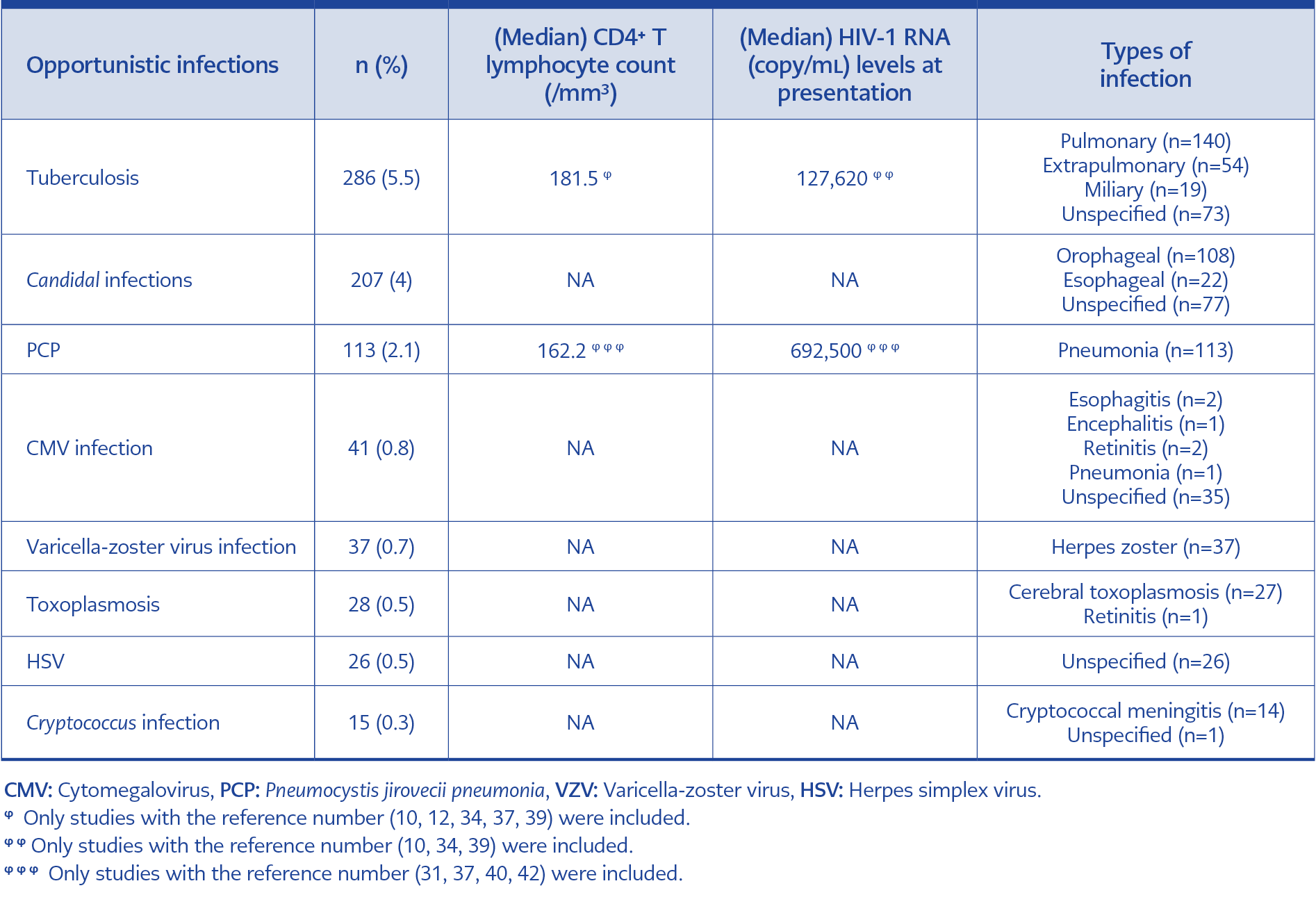
Fifty-five studies in Pubmed, 68 in WoS, and 212 in ULAKBİM TR Dizin were listed with these keywords. After removing duplicated studies, we included 22 original articles, two poster presentations, one short communication, one letter to the editor, and 17 case reports/series (Table 2). A total of 5146 patients with HIV/AIDS diagnoses were investigated among 43 relevant studies. Opportunistic infections were diagnosed in 879 out of 5146 patients (17%) with HIV/AIDS. The most frequent opportunistic infections were tuberculosis (286, 5.5%), candidal infections (207, 4%), and Pneumocystis jirovecii pneumonia (PCP) (113, 2.1%) (Table 3). Out of 1582 patients, we defined 788 patients (49.8%) as late presenters.
Among the studies, overall mortality rates of all patients with HIV/AIDS ranged between 0.8% to 36%. In terms of tuberculosis, the overall mortality was calculated as 18% in patients with AIDS and tuberculosis (9, 10, 12, 34, 37, 39). In addition, the median CD4+ T lymphocyte count was 181.5/mm3, and the median HIV-1 RNA was 127,620 copy/mL among AIDS patients with tuberculosis (Table 3). Furthermore, the overall mortality was 32.2% in patients with AIDS and PCP (32, 36, 39, 40).
Among tuberculosis patients, the clinical forms were pulmonary (140, 49%), extrapulmonary (54, 19%), miliary (19, 7%), and unspecified (73, 25%). On the other hand, among patients with candidal infections, the clinical forms were orophageal (108, 52%), esophageal (22, 11%), and unspecified (77, 37%) (Table 3).
Discussion
Although the frequency of opportunistic infections is decreasing with the prophylactic and antiretroviral treatments in patients with HIV/AIDS, HIV-related opportunistic infections still remain as an important cause of mortality and morbidity, especially for low and middle-income countries (52). Due to poor immunological status, people living with HIV (PLWH) get an increasing number of severe illnesses called opportunistic infections. Furthermore, late-diagnosed individuals with HIV have higher mortality than those diagnosed earlier, resulting in significant costs to health systems. Therefore, screening for opportunistic infections such as tuberculosis and cryptococcus, the leading causes of death among PLWH, is crucial (53). We believe that our study is the first systematic review highlighting the opportunistic infections in HIV/AIDS patients from our country regarding the clinical and laboratory results of the patients.
An updated target of 95-95-95 was released by the Joint United Nations Programme on HIV and AIDS (UNAIDS) for a pathway to ending AIDS, which is consisted of 95% of all PLWH will know their disease, 95% of all people with HIV diagnosis will receive antiretroviral therapy (ART) and 95% of all people receiving ART will have viral suppression by 2025 (54). According to UNAIDS reports, the global percentages were 84% of PLWH who knew their disease; among those, 87% were on ART, and 90% of those on ART had achieved virological suppression at the end of 2020 (55). Gokengin reviewed the targets regarding HIV infection in our country and mentioned that almost half of the patients were late presenters because of the lack of screening strategy for target populations and detection at the time of differential diagnosis (56). Between 2011 and 2016, the predicted rate of undiagnosed infection in our country was 59% and 52%, according to the HIV-TR cohort and the Ministry of Health data, respectively (57). In addition, Mete et al. also reported a cohort from İstanbul, which represents only 13% of PLWH in Turkey, and they showed that 72% and 74% of all HIV patients were diagnosed in 2016 and 2017, respectively (58).
Several important factors affect UNAIDS’s targets, such as geographic differences, income inequality, and epidemiological diversities. Late presentation is still one of the major issues in Europe, Central Asia, and migrant populations. Many PLWH (especially older people, heterosexual men, people living in low HIV prevalence areas, and migrants) remain unaware of the disease, and the percentages of undiagnosed patients range from 20% in Denmark, Sweden, and Slovakia to as high as 50% in Poland and Latvia in Europe (2). Also, in the center of Europe, the percentage of late diagnoses was recorded as high as 52.8%, which varied across transmission categories and age groups (59).
Miranda et al. analyzed 907 HIV patients in Portugal, finding that 50.6% of the patients were late presenters (60). In addition, Belat et al. also found that the pooled prevalence of late presentation to HIV/AIDS care was 52.89% in Ethiopia, reviewing eight studies with 7568 participants (61). Kostaki et al. investigated the reasons for late presentation in Greece, and they demonstrated that older age, heterosexual transmission risk group, and more recent diagnosis were associated with increased risk for late presentation (62). Similar to these findings, in our country, Karaosmanoglu et al. showed that nearly half of the patients who presented late were elderly, less educated, married, and heterosexual patients having more HIV-related diseases on admission (63).
Similarly, our study found that the percentage of late presenters was 49.8%. We believe that the major factor affecting the development of opportunistic infections with high mortality rates is late presentation in our country. Although the majority of studies included in our review were reported in the pre-pandemic period in our study, there were also reported cases that highlight the risk of misdiagnosing opportunistic infections for SARS-CoV-2 infection; difficulties in the management of opportunistic infections in a limited time to access healthcare services represent an important issue for the late presenters (64).
Szmulik et al. investigated a total number of 1313 HIV cases newly diagnosed in Poland, and they showed that PCP and esophageal candidiasis were the most common indicator diseases among AIDS cases in 2016 (65). In addition, Ali et al. found the opportunistic infection rate as 32.3% among 167 HIV patients in Qatar. They also found the most common opportunistic illness as PCP, followed by cytomegalovirus (CMV) retinitis and tuberculosis (66). Regarding opportunistic infections in small cities like Midwest Santa Catarina, Amorim et al. analyzed the records of 143 HIV/AIDS patients from Brazil, and they found that opportunistic infections were present in 38.5% of the patients (4.9% of the patients had tuberculosis) (67). On the other hand, Chepkondol et al. investigated 500 HIV-positive patients in Kenya and found the opportunistic infection rate as high as 78.8%.
Additionally, they found the most common opportunistic infection as tuberculosis with 35%, and patients with lower CD4+ T lymphocyte counts of 0-199 and 200-349 had two times greater odds of having tuberculosis than those with CD4+ T lymphocyte count of ≥ 500 (68). Among the 18 countries, including Turkey, under the program of stopping tuberculosis in the WHO European Region, the incidence of tuberculosis was estimated as 52.5 per 100,000 people (69). Wallenfels et al. investigated 88 HIV-positive patients in the Czech Republic diagnosed with tuberculosis between 2000-2020 and found that the median CD4+ T lymphocyte count was 91.5 /mm3 at the time of tuberculosis diagnosis (70). Our study also found the most common opportunistic infection as tuberculosis (5.5%), and the median CD4+ T lymphocyte count was 181.5 /mm3.
Our study has some limitations. First of all, most of the reviewed studies were retrospective and single-center studies. Thus, none of the included studies contains specific data from big cohorts and may not reflect the real epidemiology of opportunistic infections among PLWH in Turkey. Second, the results of these studies had heterogeneities regarding the different opportunistic infections with the viral loads and CD4+ T lymphocyte counts. Thus, a limited number of studies were included with the descriptive data of the specified opportunistic infection. Third, even though we tried to include all of the published studies, including case reports, the patients with comorbidities or co-infections such as syphilis, hepatitis, or malignancies were excluded. In addition, although opportunistic infections were defined as infections that occur more frequently or severely in people with immunosuppression, some included studies reported only opportunistic infections, which were stated in AIDS-defining conditions in the CDC list (71). On the other hand, Croxford et al. recently published an update about late presentation, and they mentioned that patients with evidence of recent infection, a last negative HIV test within 12 months of diagnosis, or clinical evidence of acute infection, and also previously diagnosed patients should not be considered as late presentation (72). The vast majority of included studies had not excluded recent infections when calculating late presenters as a limitation of our study.
In conclusion, tuberculosis, candidal infections, and PCP are the most common opportunistic infections among HIV/AIDS patients in our country. Prevention of opportunistic infections involves a multidisciplinary approach that includes infectious disease specialists, primary care physicians, and social workers for support. In our country, late presentation of the patients is one of the major problems, and more screening and sensitization campaigns should be carried out, such as increasing the anonymous and free HIV testing services throughout the country and education on HIV/AIDS in schools.