Introduction
Ventriculoperitoneal shunts (VPSs) have been shown to be effective life-saving treatments for hydrocephalus. Nonetheless, the occurrence of VPS-related complications and infections necessitates close monitoring and care (1). Due to blood-brain and blood-cerebrospinal fluid (CSF) barriers, many anti-infective therapeutics have difficulty reaching effective concentrations in the central nervous system (CNS) following systemic administration. Intrathecal antibiotic therapy is advised in CNS infections caused by multidrug-resistant (MDR) bacteria when systemic treatment is inadequate (2). This article describes how to handle a critically ill patient with a VPS infection caused by colistin-resistant Klebsiella pneumoniae.
Case
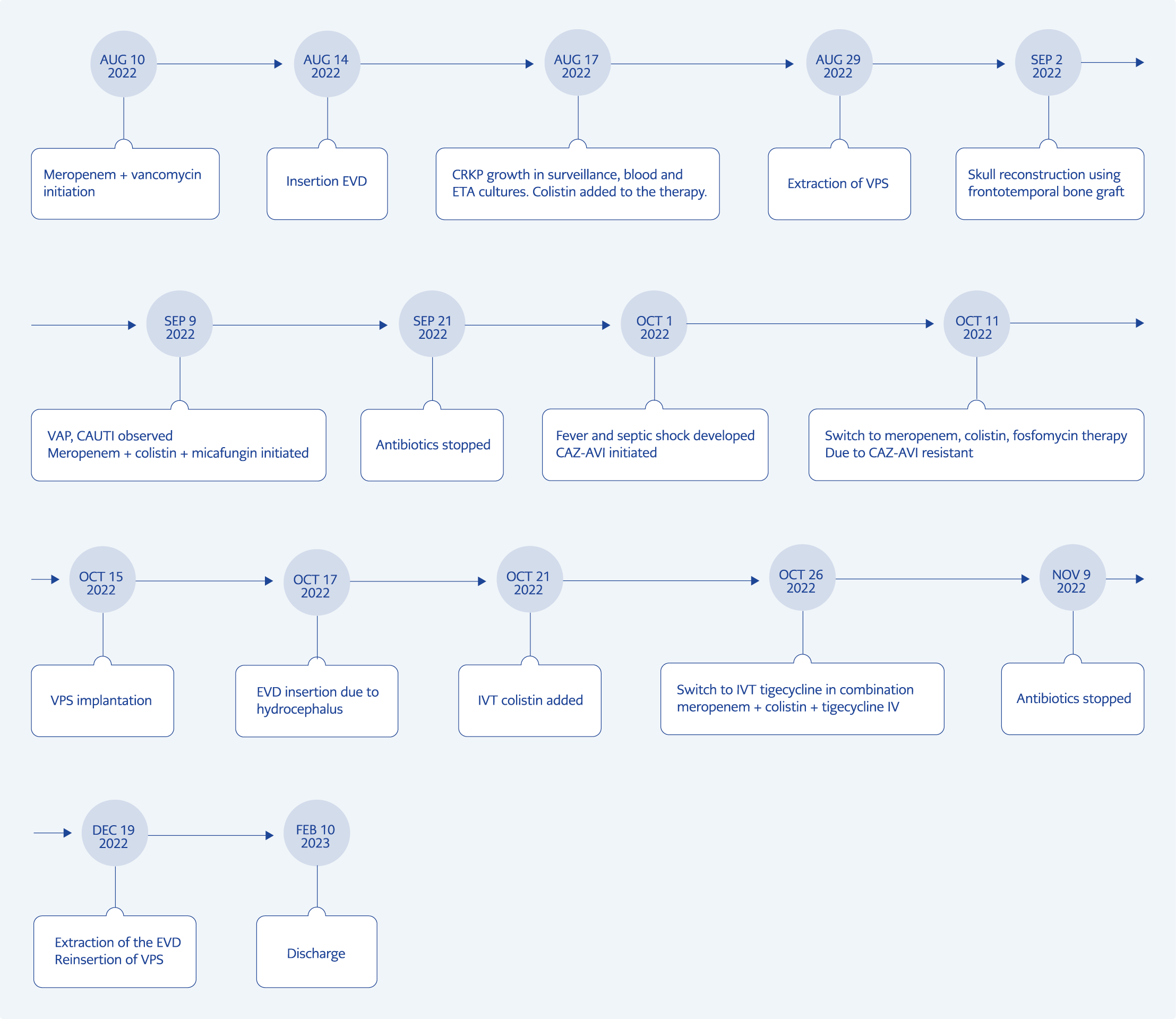
As per information gathered from the patient’s family, our 19-year-old male patient underwent surgical intervention at an external facility following a spontaneous intracranial hemorrhage, necessitating a right frontotemporal craniectomy. Subsequent to this procedure, the patient developed hydrocephalus, leading to the insertion of a VPS. The patient experienced a loss of consciousness approximately 15 days post-discharge. On August 10, 2022, the patient was urgently admitted to the emergency department of Başkent University Faculty of Medicine, exhibiting a Glasgow coma scale of 6/15, Acute Physiology and Chronic Health Evaluation (APACHE) II score of 17, and Sepsis-Related Organ Failure Assessment (SOFA) score of 6, prompting immediate intubation and admission to the intensive care unit (ICU). The patient’s body temperature was 39°C; blood pressure was 160/70 mmHg and heart rate was 190 beats/minute. Meropenem (2 g every 8 hours) in combination with vancomycin (1 g every 12 hours) intravenous (IV) therapy was initiated for the preliminary diagnosis of sepsis due to VPS-related meningitis. Upon admission, carbapenem-resistant K. pneumoniae (CRKP) was detected in the axillary, inguinal, and umbilical surveillance cultures.
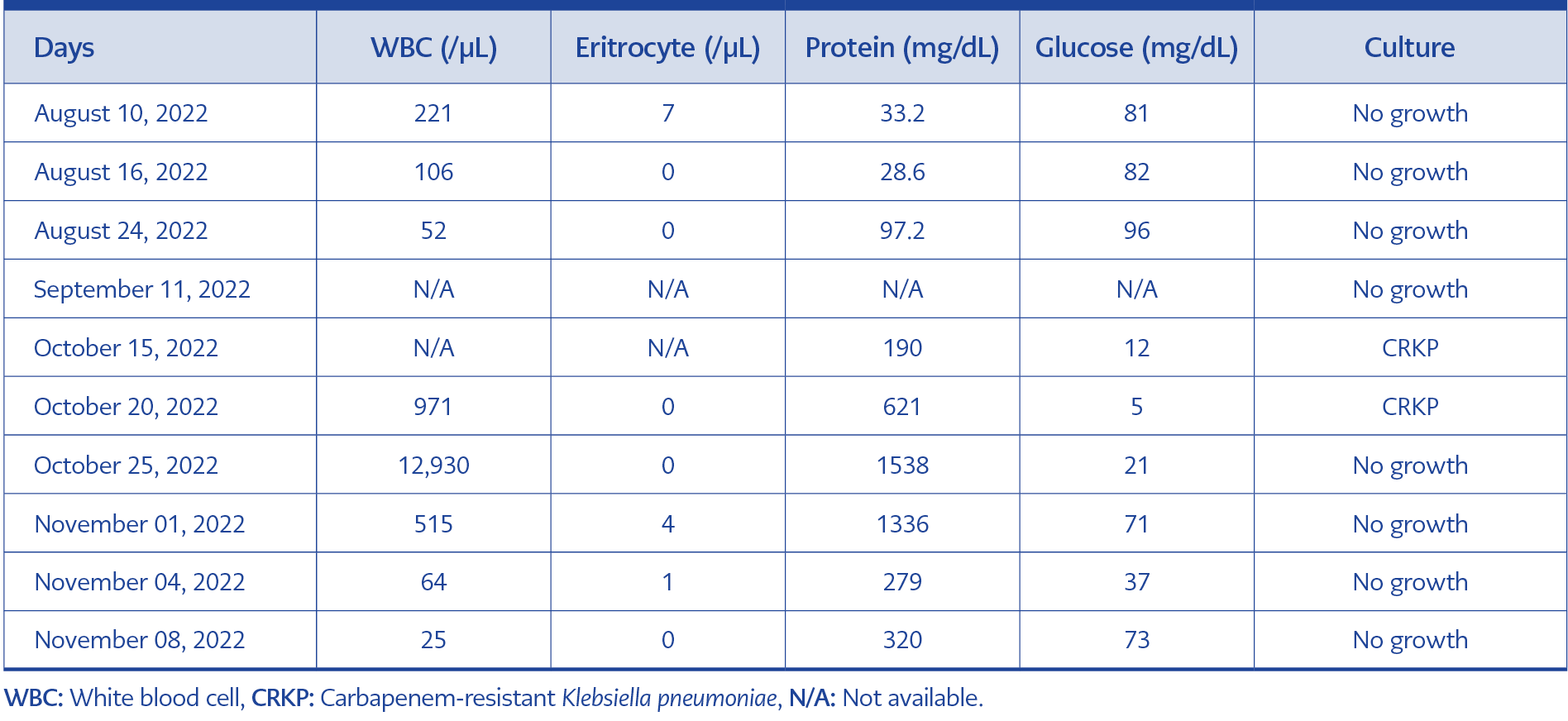
At the admission, the CSF white blood cell (WBC) count was 221/µL, the red blood cell (RBC) count was 7, the glucose level was 81 mg/dL, and the protein level was 33.2 mg/dL (August 10). The CSF test results at different times are presented in Table 1.
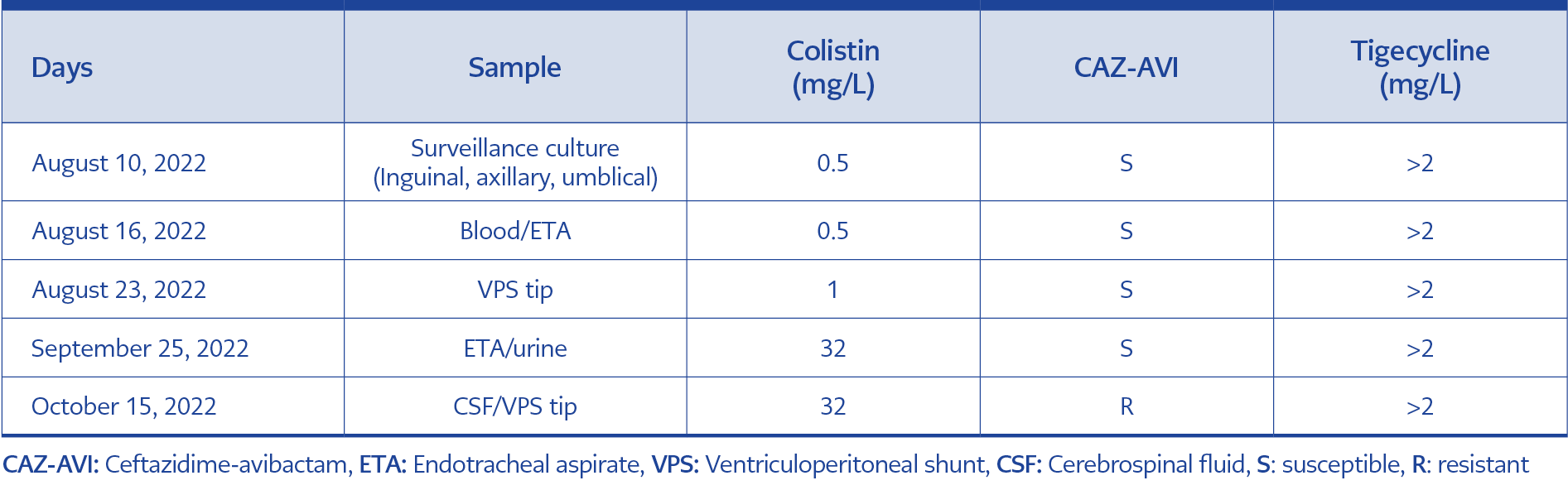
Fever and septic shock reappeared in the second month of hospitalization. Ceftazidime-avibactam (CAZ-AVI) (2.5 g every 8 hours) was initiated, and CRKP was detected in CSF cultures, as well as in blood and endotracheal aspirate cultures. However, therapy had to be switched to meropenem, colistin, and fosfomycin (4 g every 6 hours) IV combination due to the development of CAZ-AVI and colistin resistance in bacteria. K. pneumoniae antibiotic susceptibility test results on various samples are shown in Table 2. Bacterial identification and antibiotic susceptibility tests are carried out using both the conventional system and Phoenix BD automated identification and antimicrobial susceptibility testing system. Colistin susceptibility was detected using the Thermofisher SensititreTM broth microdilution method. Antibiogram data were evaluated following the recommendations of the European Committee on Antimicrobial Susceptibility Testing (EUCAST).
On October 15, the VPS was removed, and a new external ventricular drain (EVD) was inserted due to hydrocephalus observed in cranial computed tomography (CT) and shunt tip collection on abdominal CT. Three days after insertion, the patient continued to experience persistent fever (≥39 °C). We added 10 mg of colistin as an intraventricular therapy (IVT) alongside meropenem, colistin and fosfomycin IV therapy. However, the CSF values worsened after 48-96 hours of therapy. As a salvage treatment, tigecycline (loading dose 200 mg, then 100 mg every 12 hours IV and 2 mg every 12 hours one-hour lock IVT) was added to meropenem and colistin therapy (only IV therapy, IVT stopped). No side effects were observed from the IVT tigecycline administration.
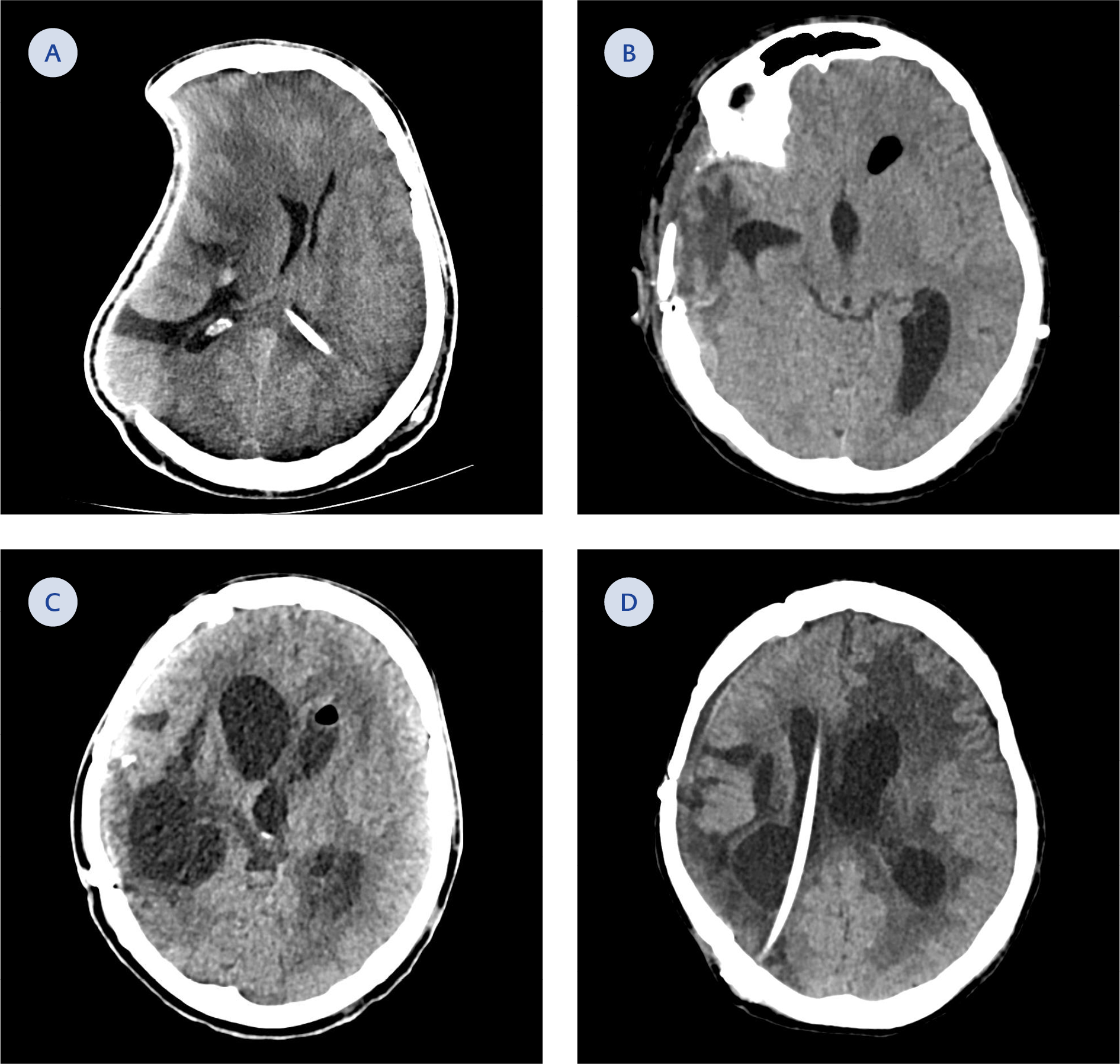
Figure 2. A Computed tomography (CT) scan of the brain. A) On day one. B) On day 47. C) On day 79. D) On day 133
Fever decreased on the seventh day of therapy. After 14 days of no growth in follow-up cultures, the ECV was removed, and a new VPS was inserted. He was discharged home from the hospital on the 185th day of ICU admission. A schematic presentation of the case is shown in Figure 1. During the hospitalization, the patient underwent a series of seven surgeries. These surgical interventions have been presented sequentially, corresponding to the patient’s cranial tomographies (Figure 2). The patient’s father signed informed consent for the publication of this case report and any accompanying images.
Discussion
This report describes the successful outcome achieved through intensive efforts in a challenging patient. Treatment for VPS infection is recognized as challenging, particularly when the causative agent is resistant to all available antibiotics in our country. As a salvage therapy, a combination of IV and IVT tigecycline with IV meropenem and IV colistin was administered, resulting in a cure.
Treatment options for CRKP are limited and vary by country. CAZ-AVI was initiated in the present case for sepsis due to CRKP. However, as indicated by recent studies, we observed rapid development of resistance to CAZ-AVI and subsequently changed our therapy to meropenem and colistin (3, 4). When CRKP was detected in CSF, meropenem IV and colistin IV/IVT were administered. However, clinical and laboratory markers deteriorated. As a salvage therapy, we added tigecycline IV/IVT to the existing meropenem plus colistin IV combination. Multiple antibiotic combinations were employed in this young patient as it was resistant to all available antibiotics. Although many guidelines advocate the administration of antibiotics to which the pathogens are sensitive, many clinicians continue to choose combinations. Despite tigecycline resistance, some research found that tigecycline and colistin worked synergistically (5, 6).
Another individualized decision is the duration of treatment, which depends on various factors, including clinical circumstances, the pathogen, the presence of a foreign body, and the ability to remove the foreign body. In uncomplicated cases, intrathecal anti-infective therapy is discontinued 48 to 72 hours after the CSF culture turns sterile (7). In contrast, for patients with repeated positive CSF cultures despite successful anti-infective therapy, the Infectious Diseases Society of America (IDSA) recommends continuing treatment for 10 to 14 days after the last positive CSF culture (8). Curebal et al. noticed that the combination of IV tigecycline + IV amikacin failed to treat an 8-month-old infant with VPS infection caused by extremely drug-resistant (XDR) K. pneumoniae. IVT was replaced with tigecycline, and the patient was cured after 14 days (9). Wang et al. found no clinical improvement with cefoperazone-sulbactam plus polymyxin B (IV/IVT) or meropenem plus CAZ-AVI plus fosfomycin combination therapies for XDR K. pneumoniae intracranial infection. However, as salvage therapy, the combination of tigecycline (IVT) plus meropenem plus CAZ-AVI in the patient achieved success in 10 days (10). In line with previous studies, changes in treatment duration, antibiotic combinations, and IVT therapy dosages have been described.
The present report had some limitations. Neither CSF nor blood could be demonstrated to have reached effective antimicrobial concentrations. The size of the ventricles, basal cisterns, and subarachnoid space above the convexities and in the spinal canal all influence the volume of CSF space in individuals. Combination therapy was administered despite in vitro resistance to all available antibiotics.
In conclusion, combining IVT and systemic tigecycline therapy may be effective as rescue therapy for CNS infections caused by CRKP. The use of intraventricular tigecycline in the treatment of CRKP appears promising; however, appropriate dosage adjustments and suggestions based on evidence are required.