Introduction
The presence of cognitive disorders in the human immunodeficiency virus (HIV) infected individuals has been known for a long time. A complete neuropsychological evaluation method is a gold standard for diagnosing HIV-related cognitive impairment (HAND). However, such tests require specially trained personnel and specialized centers, and testing takes a long time (1). Montreal Cognitive Assessment (MoCA) is a test originally developed to screen for cognitive impairment in the geriatric population at risk of early dementia. MoCA tests a wide range of cognitive domains, including memory, attention, and frontal executive functions, which are often affected in HIV-infected patients. Accordingly, it is sensitive to detecting mild cognitive impairments. Also, it can be applied both for baseline and follow-up assessments (2, 3).
The use of antiretroviral therapy (ART) has resulted in a significant decrease in HAND incidences (4, 5). A combination of antiretrovirals, which have better penetration through the blood-brain barrier, might be more effective in the treatment of HIV patients (6).
Our study aimed to evaluate the cognitive functions of HIV-infected patients in our clinic through the MoCA test, determine the factors affecting cognitive functions, and compare the cognitive functions before and after the treatment.
Materials and Methods
The study was conducted prospectively between January – June 2017 in Şişli Hamidiye Etfal Training and Research Hospital. Fifty-two HIV-positive patients were included in the study. Inclusion criteria were being between the ages of 20-35, being diagnosed with HIV recently, and planning to start treatment. Exclusion criteria were having neurological or psychiatric comorbid disease, systemic infection, metabolic disorder, anemia, hypo-hyperthyroidism, positive syphilis serology, history of substance or alcohol abuse, head trauma, or central nervous system infection. Beck Depression Inventory (BDI) was used to exclude patients with depression. Patients with more than nine points were excluded because of possible depressive states. The study was reviewed and approved by the Ethics Committee of Şişli Hamidiye Etfal Training and Research Hospital in Istanbul, Turkey.
The patients with eligible criteria were evaluated just before the initiation of the treatment. Montreal Cognitive Assessment Scale (MoCA) was used to evaluate cognitive functions. The same physician performed it, and cognitive impairment was evaluated by using the suggested cut-off score of ≤ 21 for our country (7). The patient’s age, gender, education level, smoking habit, alcohol and substance use, cluster differentiation 4 (CD4) + T lymphocyte count, HIV RNA level, and toxoplasma serology were recorded, and they were classified according to these parameters. In order to determine the effect of education on the MoCA test, patients were divided into two groups, higher education graduates and non-higher education graduates. Also, they were divided into groups by levels of CD4 + T lymphocyte (over 500 cells/uL or below) and HIV RNA (over 104 copies/ml and below) to show their effects on MoCA scores. Finally, MoCA scores of Toxoplasma IgG (+) and IgG (-) groups were compared to assess the effects of previous toxoplasma infection on cognitive functions.
Statistical Analysis
Average, standard deviation, median, lowest, highest, frequency, and ratio values were used in the data’s descriptive statistics. The distribution of variables was measured by the Kolmogorov-Smirnov test. Mann-Whitney U test was used in the analysis of independent quantitative data. Wilcoxon test was used to analyze dependent quantitative data. Spearman’s correlation analysis was used in the correlation analysis. P-values were corrected for multiplicity.
The statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS) 22.0 (IBM Corp., Armonk, NY, USA).
Results
A total of 52 patients were included in the study. Two female patients were excluded from the study because of the possibility of affecting the sample distribution. Patients with elevated BDI, positivity in syphilis serology, and diagnosed with bipolar disorder were also excluded. The study continued with a total of 40 patients.
All patients were male, and the mean age was 29.1±4.0 (age range= 25-35). The patients’ baseline median CD4 + T lymphocyte count was determined as 515.0 (interquartile range [IQR]=374) and baseline median HIV RNA level determined as 75,100 copies/ml (interquartile range [IQR]= 207.850).
The patients whose treatment was started and who had treatment compliance were re-evaluated at the sixth month of the treatment. Depressive symptoms were evaluated again with the BDI, and MoCA test was performed. Follow-up CD4 + T lymphocyte and HIV RNA levels were recorded. MoCA test results of the patients were compared with their pre-treatment values. In addition, MoCA scores of viral suppressed and non-suppressed groups were compared, and the changes in MoCA scores in these two groups were examined.
MoCA Test
The MoCA test (Turkish version 7.1) evaluates attention, concentration, executive functions, memory, language, visual structuring skills, abstract thinking, calculation, and orientation. The application duration of the test is approximately 10 minutes. The highest score obtained from the test can be 30 (7).
Baseline Assessment
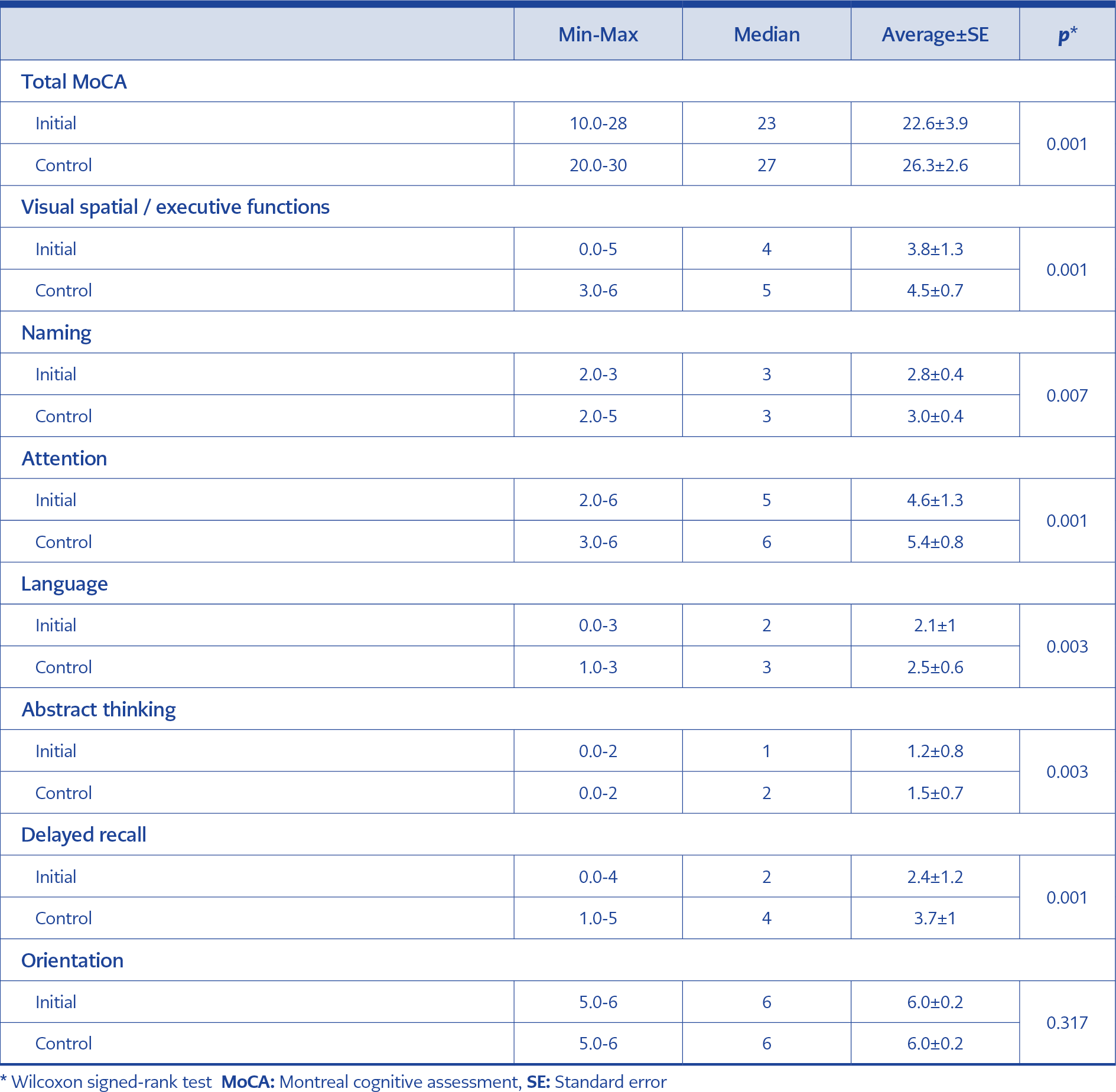
The mean baseline MoCA score of the patients was 22.6±3.9. It was below the normal value in 11 (27.5%) patients, and the most affected cognitive functions were delayed recall and attention, respectively.
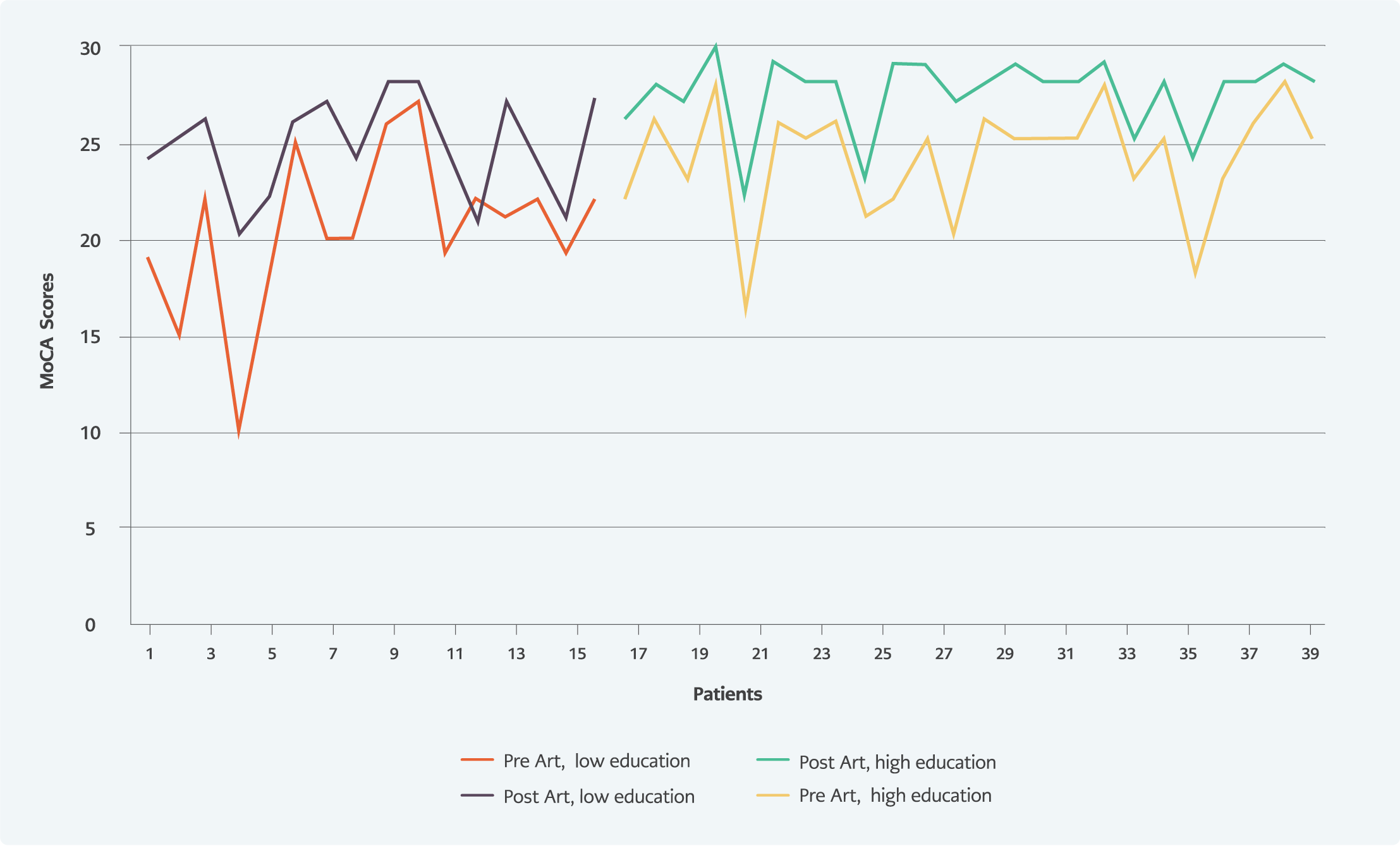
The baseline MoCA score was significantly higher in the group of higher education graduates than in non-higher education graduates (p<0.05) (Figure 1). MoCA visual-spatial / executive functions score, MoCA attention score, MoCA abstract thinking score; the subgroups of MoCA, were significantly higher in the higher education group. No relationship was found between smoking, substance or alcohol use, and the MoCA score.
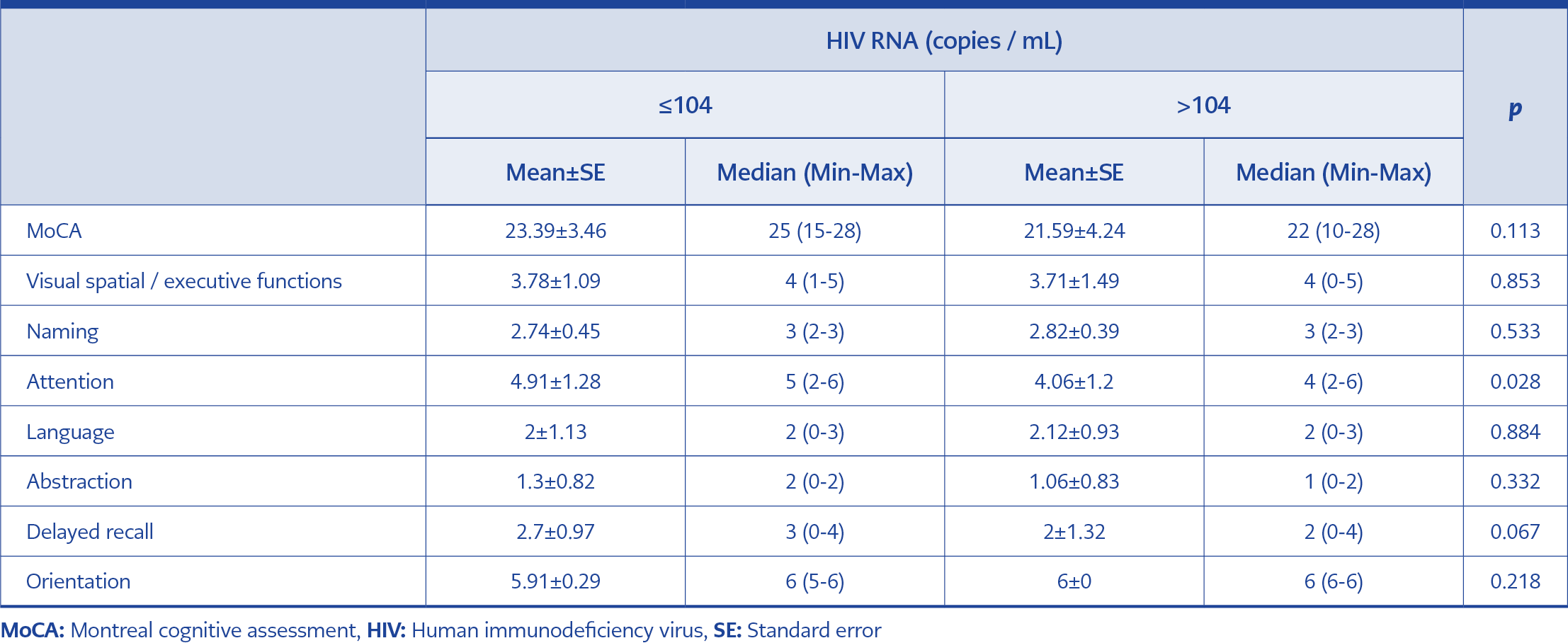
There was no significant difference between the total MoCA score of the high (42.5%) or low HIV RNA level group (57.5%). However, the low HIV RNA level group’s attention score was significantly higher than the high HIV RNA level group (p<0.05). The relationship between the baseline HIV RNA and total MoCA scores and MoCA subgroup scores is presented in Table 1.
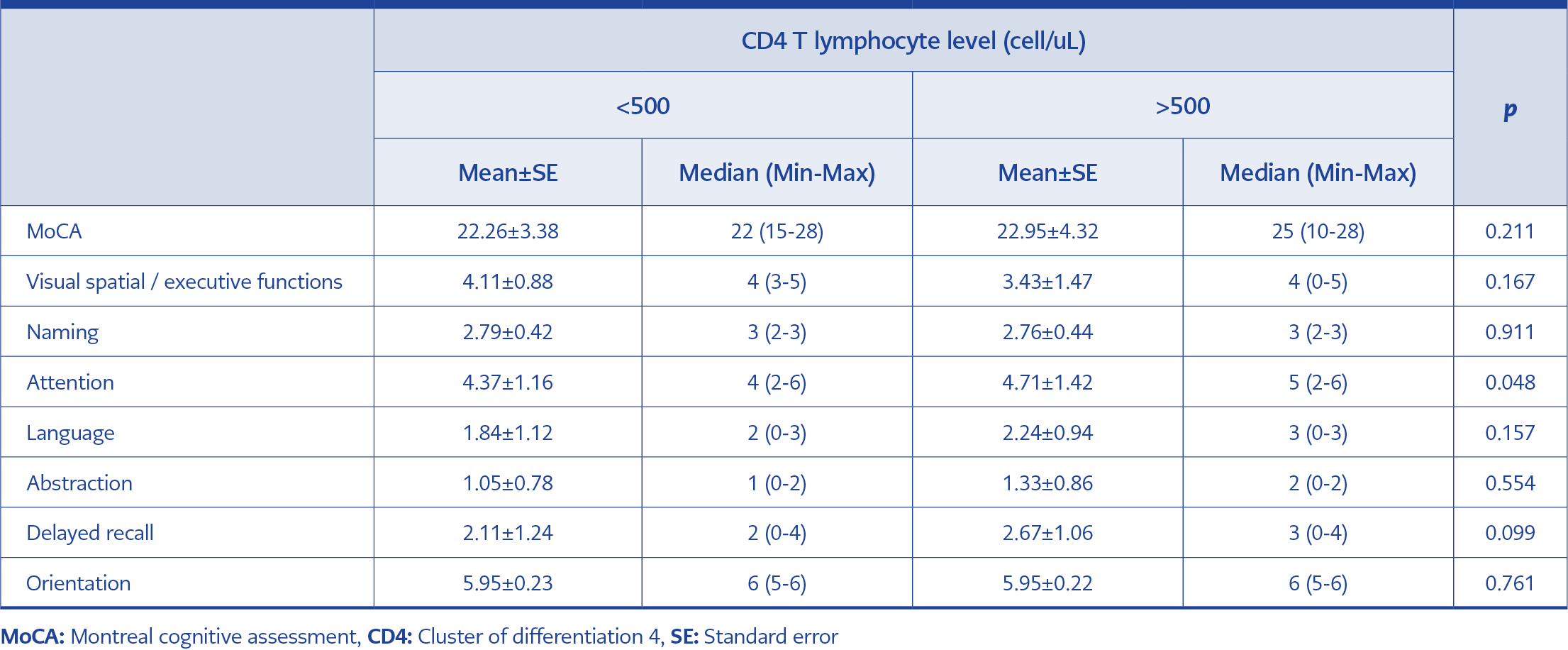
When the groups were compared according to the CD4 + T lymphocyte levels, no significant difference was found in the total MoCA score. However, the attention score was significantly higher in the group with a high CD4 + T lymphocyte (52.5%) level than in the group with a low CD4 + T lymphocyte (47.5%) level (Table 2).
Ten patients included in the study had a toxoplasma infection history. When the groups with negative and positive toxoplasma serology were compared, no significant difference was found regarding cognitive functions.
Follow-up Assessment
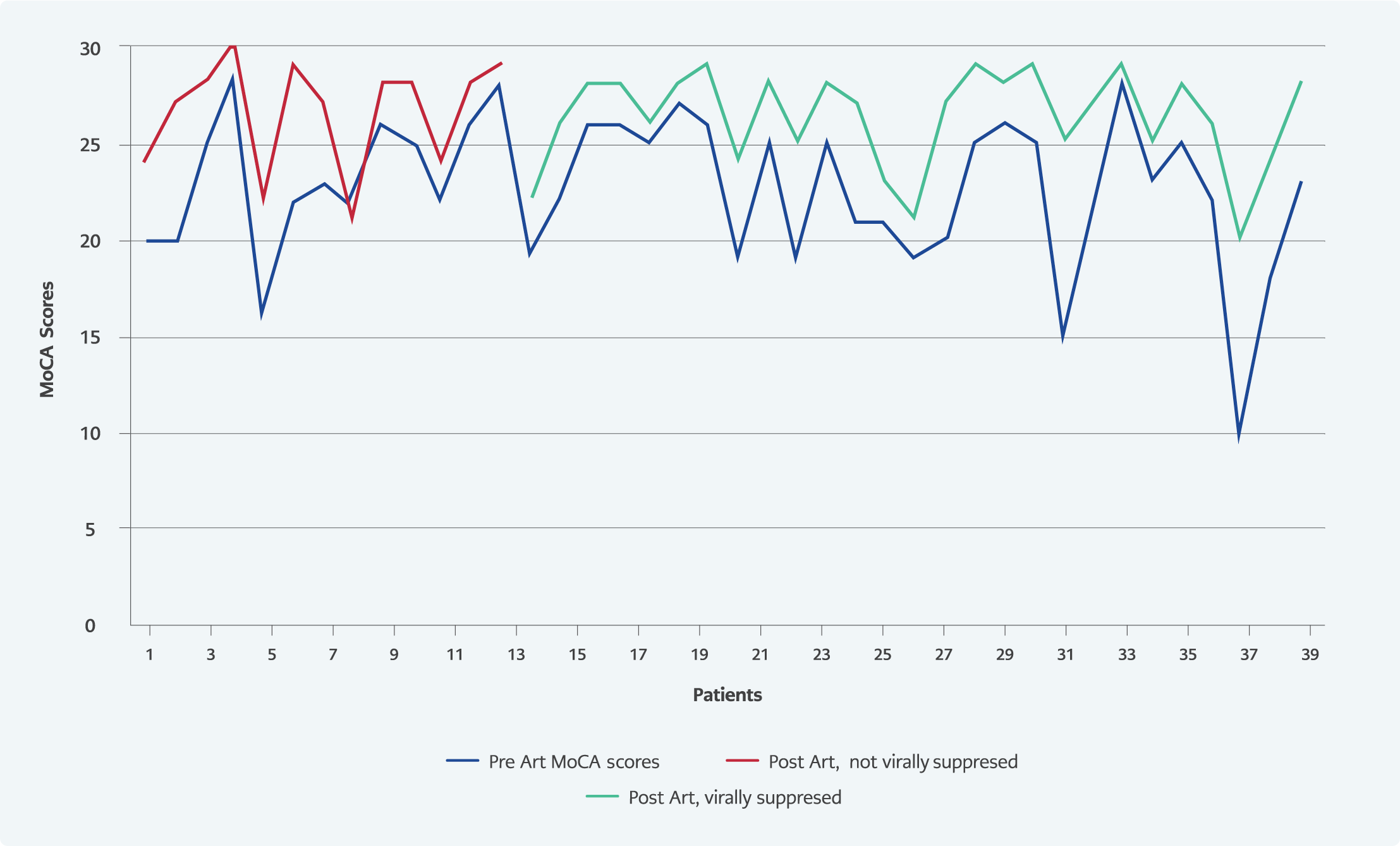
Follow-up MoCA score increased significantly compared to the baseline MoCA score (p<0.05). MoCA score was found below the normal value in 3 (7.5%) patients in the sixth month of the treatment. When the changes in MoCA subgroups were examined, it was observed that there was an improvement in all cognitive functions except the orientation function (Table 3, Figure 1).
There was no significant difference between the MoCA scores of the patients whose HIV RNA fell below 50 copies/ml and those whom HV RNA did not fall below the threshold. Patients were also examined in terms of changes in MoCA scores. The change in the total MoCA score and delayed recall subgroup score of the group whose HIV RNA level was below 50 copies/ml was significantly higher than the group whose HIV RNA level did not fall below 50 copies/ml (p<0.05).
When the two groups that CD4 + T lymphocyte counts below and above 500 cells/uL were compared, no significant difference was found between the follow-up MoCA scores.
Discussion
HIV, which is known to have effects on many systems of the body, especially the immune system, also affects the central nervous system from the early stages of the disease. Cognitive impairment caused by the virus may vary clinically from asymptomatic to severe cases such as dementia.
Many studies have supported the positive effect of ART on cognitive functions. It has been reported that ART usage is particularly effective in the improvement of attention functions, verbal rationality, and visual and executive functions (8-12).
In our study, we used the MoCA test to assess neurocognitive impairment. The most impacted cognitive functions were delayed recall and attention functions. The numbers of patients with cognitive disorders before and after the treatment were 11 (27.5%) and 3 (7.5%), respectively. After the treatment, improvements were observed in the patients’ cognitive functions, including visual/spatial executive functions, naming, language, abstract thinking, delayed recall, and attention functions. Significant improvement in MoCA scores after ART indicates that ART had positive effects on cognitive functions, as shown in the literature.
Differences in educational experiences may result in lower performance on neuropsychological tests. For example, a study found cognitive impairment at 17% in HIV-positive patients with high school and higher education levels and 38% in others who received education below high school (13). Education level also affects MoCA scores (14). In our study, MoCA scores were assessed according to the education level, and the scores were significantly higher in the university graduates group.
Studies show that smoking affects cognitive functions, especially learning and memory (15, 16). Alcohol use in HIV-infected patients causes rapid progression of HIV infection and increases HIV’s negative effects on cognitive functions, especially executive functions and reaction time (15, 17). Our study found no relation between smoking, alcohol use, and cognitive functions.
Regarding the relationship between HIV-related conditions and cognitive functions, studies show that a low CD4 count is one of the most important factors (18, 19). In our study, there was no significant difference between the low and high CD4 count level groups in terms of total MoCA scores, but the attention scores were significantly higher in the high CD4 count level group.
Different results were found in studies examining the relationship between plasma HIV RNA levels and cognitive functions. In a study including 140 patients in the United States of America, patients were divided into three classes according to their viral loads, and their cognitive functions were compared by neuropsychological tests. There was no significant difference between these three groups regarding cognitive functions (20). A study from Australia applied neuropsychological tests to groups with and without viral suppression and found no significant difference between the two groups concerning cognitive functions (21). In another study, HIV RNA values above 4.5 log 10 copies were found to be associated with an increased risk of cognitive dysfunction (22). Our study found no significant difference in the MoCA tests of the groups separated according to HIV RNA level, but MoCA subgroup scores measuring attention were significantly higher in the low viral load group. In the sixth month of treatment, it was seen that viral suppression did not make a significant difference in terms of cognitive functions. However, the change in the total MoCA score (difference in the follow-up baseline MoCA score) and the delayed recall subgroup score in the group with viral suppression were significantly greater than in the group without viral suppression (Figure 2).
Although virological suppression has been shown to be important, the degree of cognitive impairment may affect drug adherence (23). If patients do not take their ART, they cannot achieve virologic suppression resulting in having HAND; because of this, they can forget to take their ART. This results in a vicious circle. Therefore, starting treatment earlier and ensuring drug adherence are very important.
Studies conducted with the MoCA test in HIV-positive patients have reported that this test was promising for the early detection of cognitive functions and in long-term clinical follow-up (24). In a study in which the MoCA test was used to diagnose HAND in HIV-positive patients over 60 years old, the test’s sensitivity and specificity were 72% and 67%, respectively (25). The sensitivity and specificity of the test may vary depending on the cut-off value determined for the test. Overton et al. reported a prevalence of HAND of 64% by using MoCA in 119 HIV-infected individuals. MoCA had a sensitivity of 59% and a specificity of 81% by using a cut-off point as <26, and the sensitivity increased to 83% if the cut-off point was taken as <27 (26). Optimal cut-offs for the MoCA test vary by race, education, and ethnicity. In a study conducted in Malaysia, demographically corrected MoCA scores were used to determine the prevalence of HAND, and demographically corrected rates were in agreement with international HAND prevalence rates in treated and virally suppressed HIV-positive populations (27). In our study, we used the Turkish version of the MoCA test, which was validated for Turkey, and determined the cut-off value of the test as 21, which is commended value for our country (7, 28, 29).
Our study has some limitations. The first one is the small number of patients, and the second is the effect of educational status on the MoCA test.
In conclusion, the MoCA test is recommended as an alternative for screening and follow-up purposes in HIV-infected patients because it is sensitive to mild cognitive impairment and can monitor the cognitive functions frequently affected by HIV. Our study supports that the MoCA test can be used for monitoring cognitive functions in patients receiving treatment. Besides, evaluating patients’ cognitive functions before the treatment is important for treatment selection and clinical follow-up. Early initiation of appropriate ART seems the best solution for preventing and treating cognitive dysfunctions.