Introduction
Antimicrobial resistance (AMR) is declared one of humanity’s top ten global public health threats (1). AMR is associated with increased hospitalizations, extended hospital stays, readmissions, and a higher mortality risk (2). In 2019, 4.95 million deaths, including 1.27 million directly attributable, were associated with bacterial AMR. Sub-Saharan Africa has the highest all-age death rate attributable to AMR at 27.3 deaths per 100,000 (3). In 2015 and 2017, the WHO and Tanzania Mainland Ministry of Health launched the Global and National Action Plans for AMR with five goals: raising awareness and sharing accurate information, improving surveillance and research, reducing infections, enhancing the proper use of antimicrobial agents, and creating a strong economic rationale for sustainable investments. In 2023, Tanzania added a sixth goal to improve coordination and governance in combating antimicrobial resistance (4-6).
Irrational drug use, particularly antimicrobials, is the primary drive accelerating the increase of AMR (7, 8). In low- and middle-income countries, there has been a reported significant rise in antimicrobial use (AMU) and consumption in recent decades (9). Regular AMU surveillance improves knowledge and prescription practice among healthcare providers, fostering advanced, rational prescribing and use of antimicrobials (10). Thus, AMU is identified as a particularly indispensable target for surveillance interventions (11). Several tools have been developed to serve this purpose. It is essential to survey prescribing tendencies and the use of antimicrobials in outpatient and emergency care departments because they serve many patients, and most prescriptions happen there (12).
The World Health Organization/International Network of Rational Use of Drugs (WHO/INRUD) indicators are surveillance tools used to understand the pattern of AMU in outpatients (13). It measures prescribing performances for health facilities. Several studies assessed AMU using the WHO/INRUD indicators. A study in Kenya, an East African country, conducted AMU surveillance using the WHO/INRUD indicators from rural counties in ten public primary healthcare centers. Of all prescriptions assessed, 84.8% had antimicrobials prescribed higher than the recommendation by WHO (20.0-26.8%). Prescription of injection was 24.9% (recommended 13.4-24.1%), and the almost optimal percentage of prescribed drugs from the Kenya Essential Medicines List was 96.7% (recommended 100%) (14). In Tanzania, several studies have assessed AMU, antimicrobial consumption (AMC), and AMR, leading to interventions and establishing antimicrobial stewardship teams; however, limited data is available on prescribing antibiotics in Tanzanian hospitals, especially in tertiary hospitals for outpatients. Even though outpatients represent about 90% of all AMU, with more than half of these prescriptions being either unnecessary or inappropriate (12, 15) no adequate studies have been conducted on AMU using the WHO/INRUD indicators or outpatient and emergency care for tertiary zonal referral hospitals. Our study, conducted at a tertiary and zonal referral hospital in the central zone of Tanzania, aimed to analyse antibiotic prescribing practices and trends among outpatients to provide valuable insights for developing successful antimicrobial stewardship initiatives using the WHO/INRUD indicators.
Materials and Methods
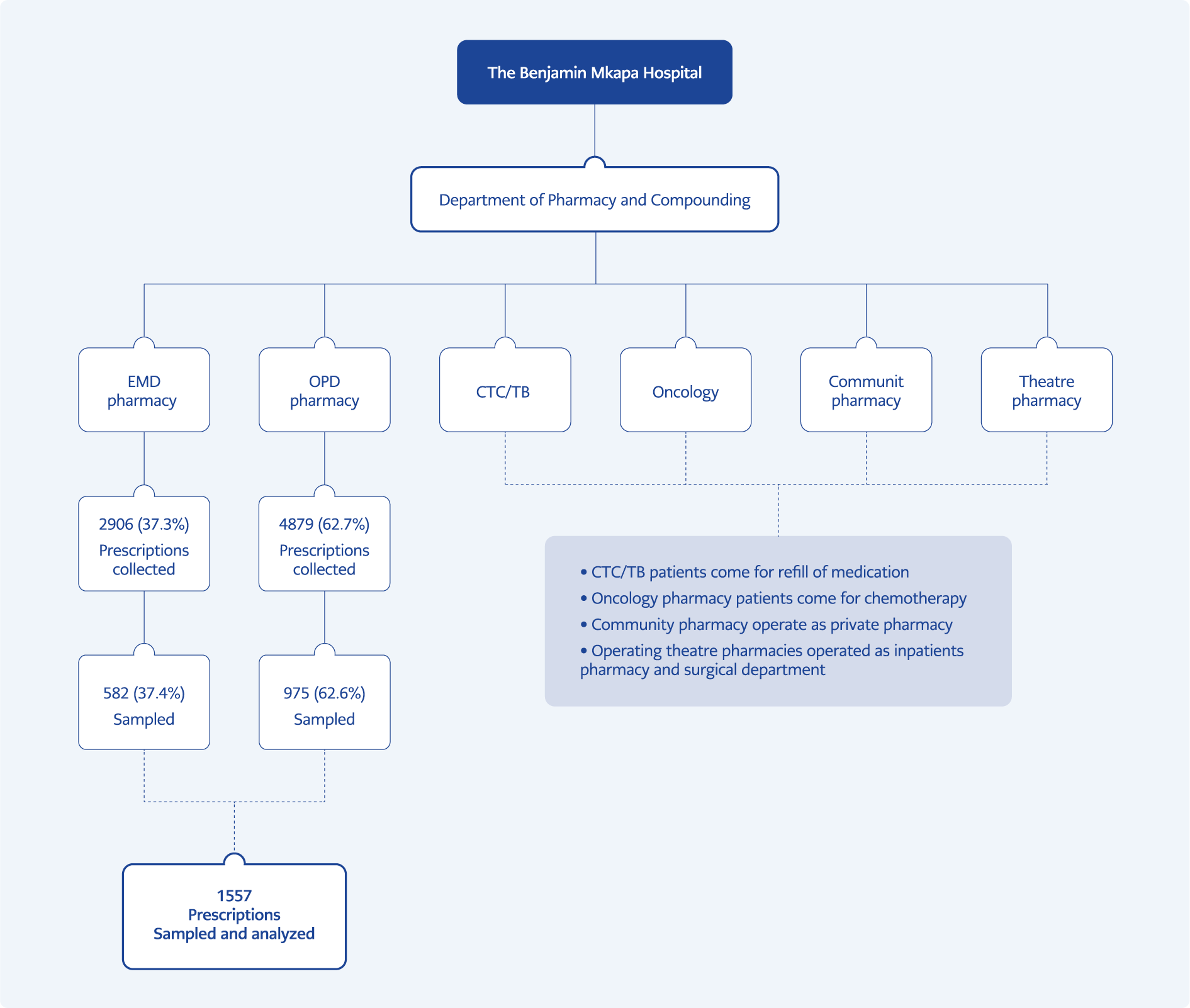
This prospective descriptive study was conducted at Benjamin Mkapa Hospital (BMH), a tertiary and zonal referral hospital in the central zone of Tanzania, with nearly 400 beds capacity. AMU data for outpatients were collected from August 01 to October 31, 2022, using the WHO/INRUD indicators to investigate drug use in health facilities (13). Data were collected prospectively daily for three consecutive months (August, September and October). The simple random sampling method was used to select the samples, and every fifth prescription was selected in chronological order to obtain at least 600 prescriptions from the outpatients’ clinics and emergency department pharmacies as advised by the WHO/INRUD indicators (13). The variables of this study were determined according to the WHO/INRUD indicators. Demographic variables, such as age and sex, were also collected. The indicators presented in Supplement 1 were used to assess AMU in outpatients.
AMU data from pharmacies in the outpatient department (OPD) and emergency department (EMD) were collected. We excluded all inpatient prescriptions. The statistical analyses were performed using the Statistical Package for Social Sciences (SPSS) 20.0 (IBM Corp., Armonk, NY, USA). Tables and chats were used in data presentation to disseminate and publish data on AMU.
The National Institute for Medical Research and the National Health Research Ethics Review Committee (NatHREC) granted the study an ethical clearance certificate with reference number NIMR/HQ/R. 8a/Vol. IX/4260. The decision date was March 31, 2023, and the BMH authority permitted the research to be conducted in the hospital.
Results
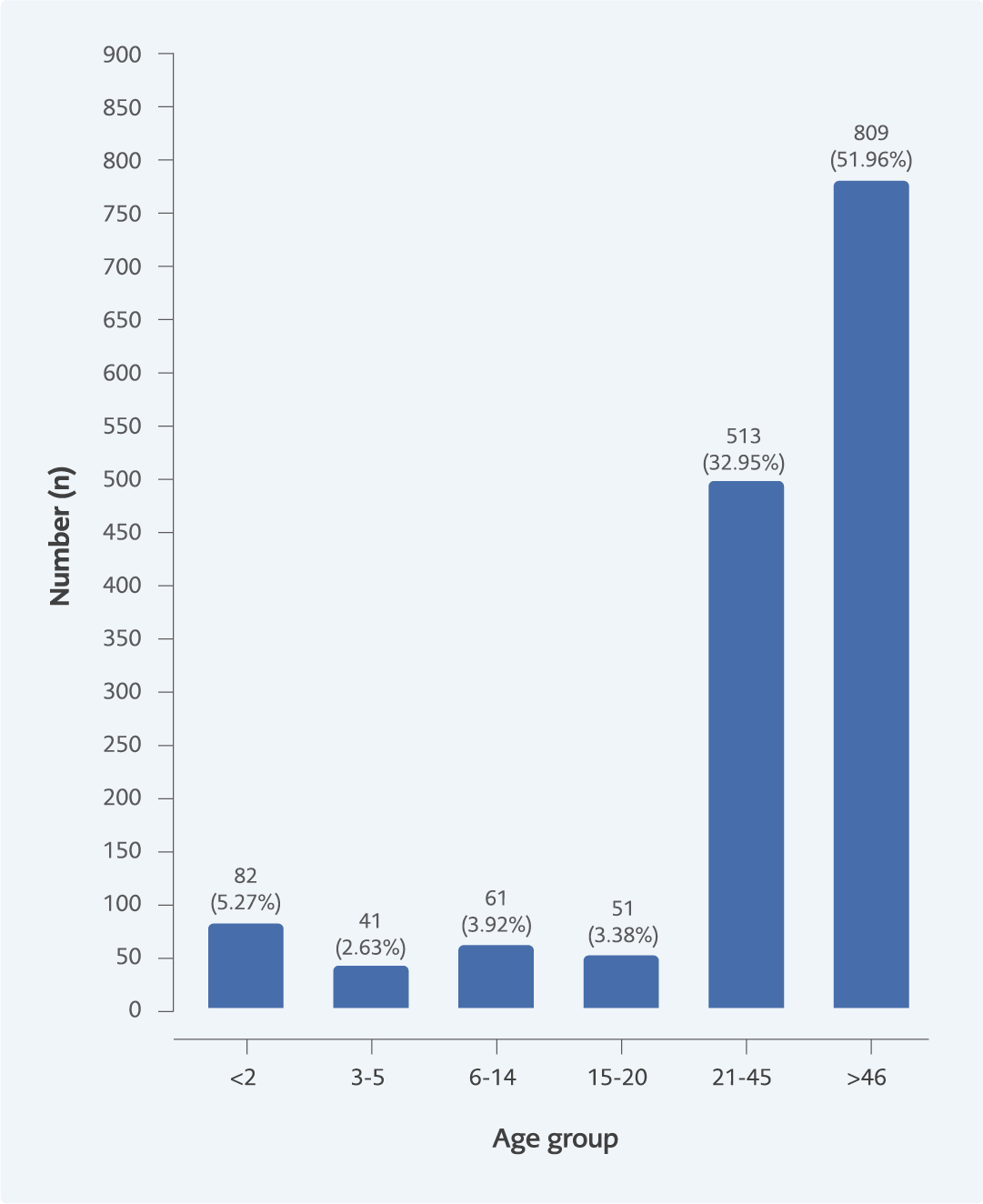
A total of 1557 prescriptions were sampled and analyzed. Of these, 975 (62.6%) were from OPD, and EMD 582 (37.4%) pharmacies (Figure 1). Most patients who attended OPD and EMD pharmacies were above 40 years old (Figure 2).
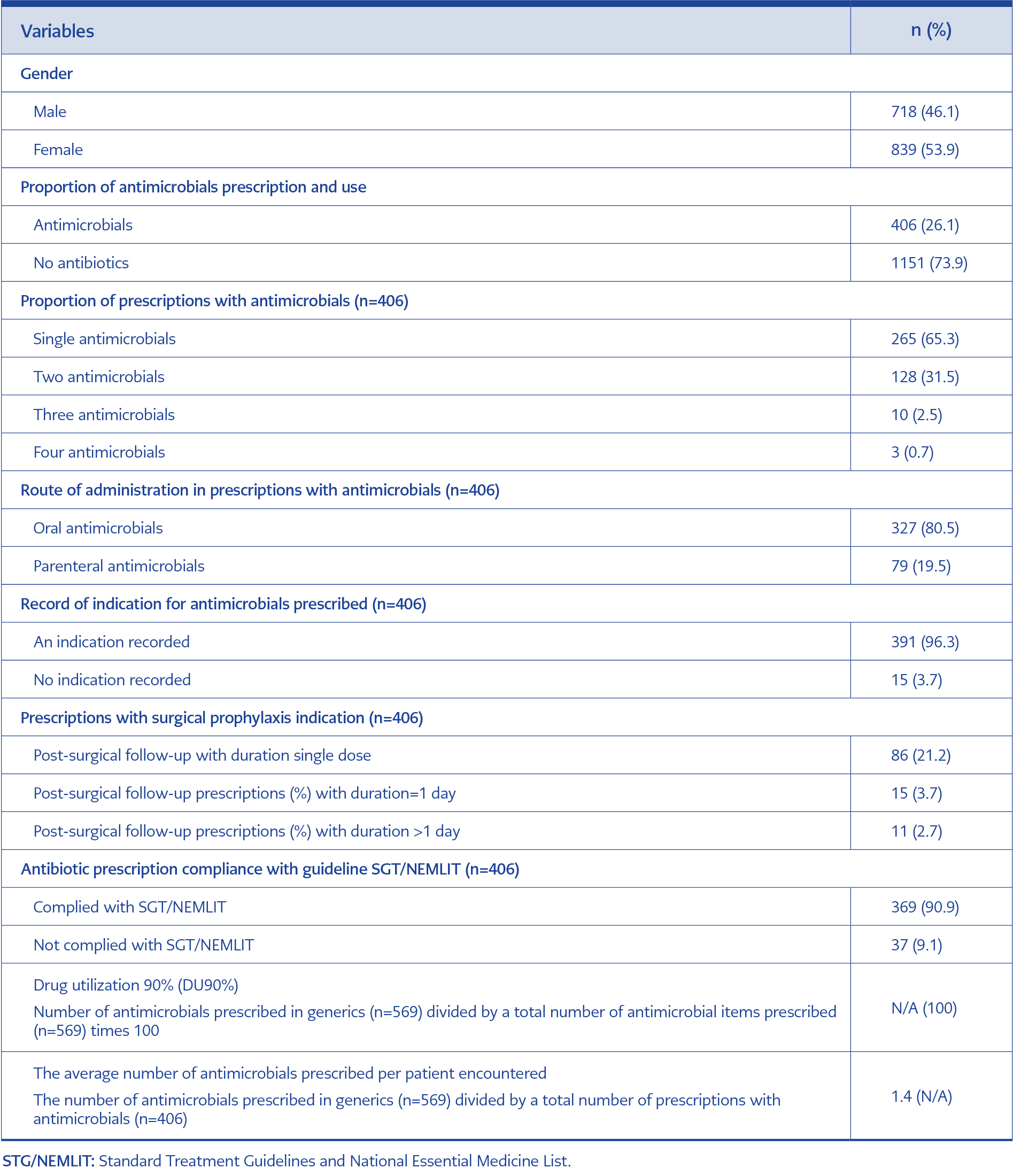
About 26.1% of surveyed prescriptions included antimicrobials (Table 1). Of these, 90.9% complied with current National Treatment Guidelines. Most antimicrobials are prescribed as a single antimicrobial, with 65.3% out of all assessed prescriptions, while about 19.5% of all prescriptions with an antimicrobial presented with parenteral. The total number of antimicrobial items prescribed was 569. The number of prescriptions with antimicrobials was 406, which implies 1.4 antimicrobials per prescription.
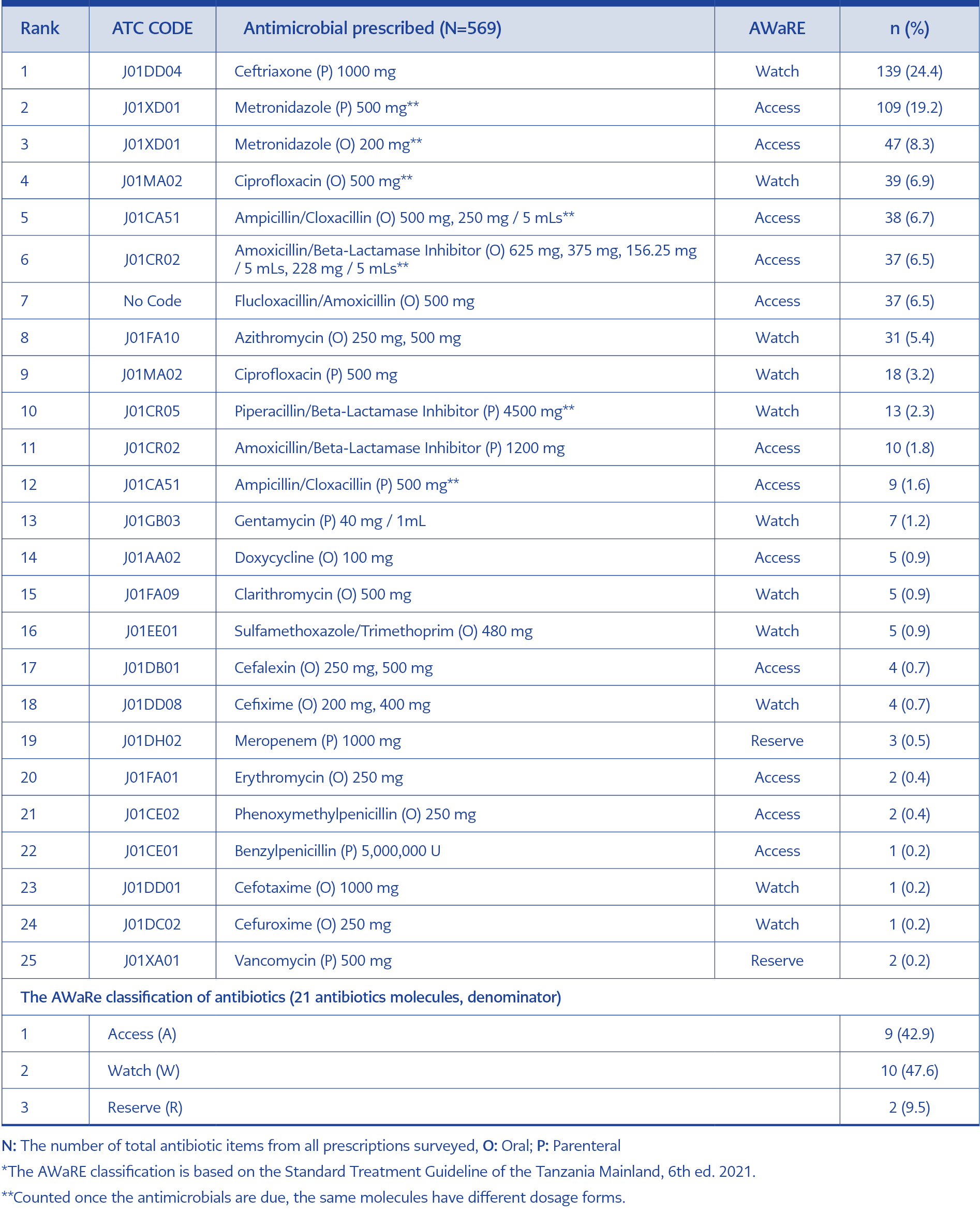
These were classified into 21 groups, according to the WHO AWaRE (Access [A], Watch [W], and Reserve [R]) classification and route of administration (Parenteral (P) and Oral [O]). We identified 9 (42.9%) antimicrobials as an A, 10 (47.6%) W, and 2 (9.5%) were R. The top five used antimicrobial were ceftriaxone (P, W) 139 (24.4%), metronidazole (P, A) 109 (19.2%), metronidazole (O, A) 47 (8.3%), ciprofloxacin (O, W) 39 (6.9%) and ampicillin/cloxacillin (O, A) 38 (6.7%). While reserved antimicrobials were used with less than 1% (Table 2). Ampicillin-cloxacillin fixed-dose combination (FDC) (O) and flucloxacillin-amoxicillin FDC (O) were highly prescribed antimicrobial combinations.
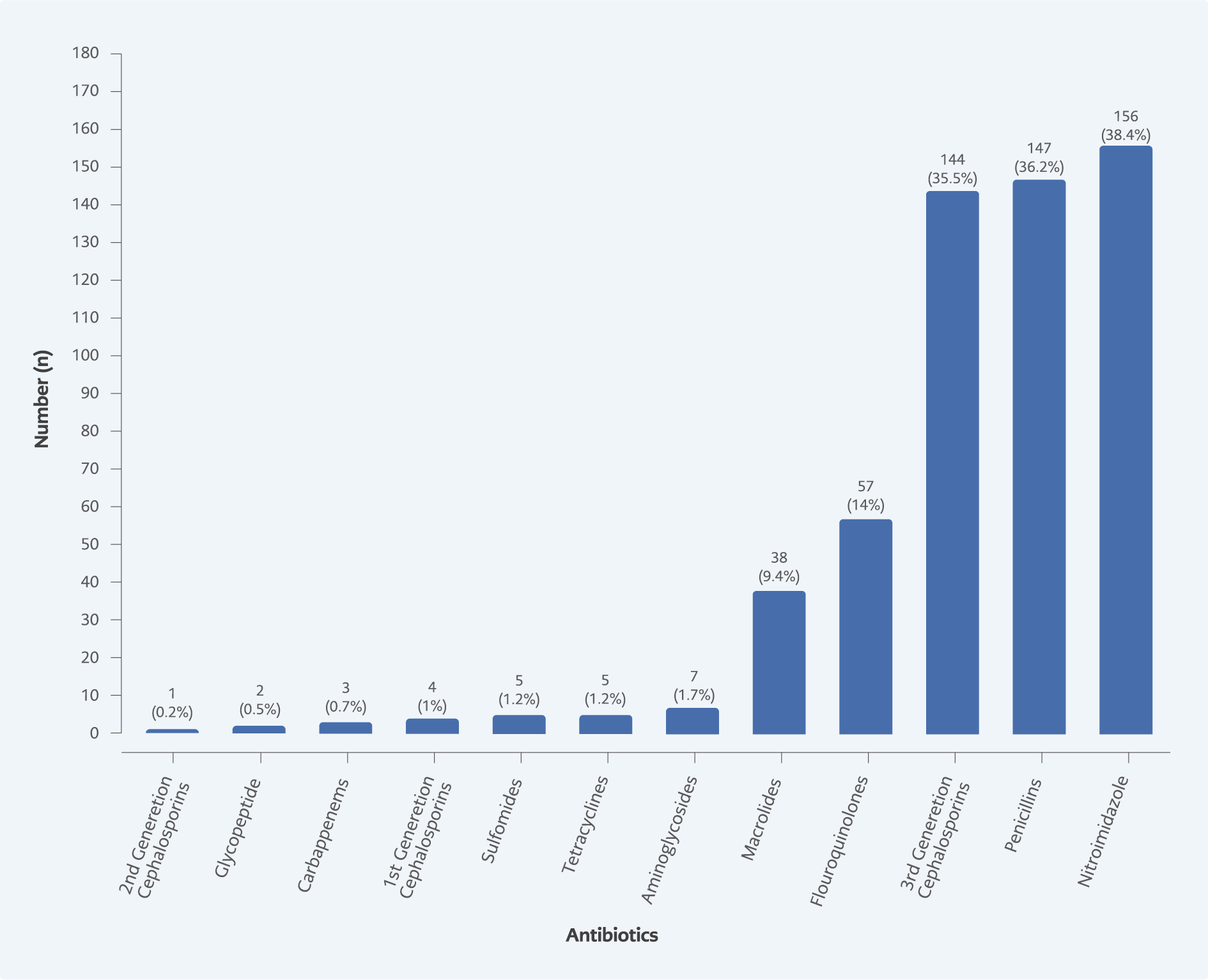
Results from this study indicated nitroimidazole derivatives were highly prescribed at 156 (38.4%), followed by penicillin and third-generation cephalosporin at 147 (36.2%) and 144 (35.5%), respectively. In contrast, aminoglycoside, sulfonamide, tetracycline, first-generation cephalosporin, glycopeptide, second-generation cephalosporin, and carbapenems are the least prescribed antimicrobials, which occupied less than 1% of use (Figure 3).
The most typical indications were post-surgical follow-up and urinary tract infection, with 112 (27.6%) and 52 (12.8%), respectively; cough was next at 38 (9.4%), while septicemia and soft tissue infections had 35 (8.6%) (Table 3). The surveillance assumed that each prescription had only one indication.
Discussion
Our study found that BMH has achieved 100% drug utilization with all prescribed antimicrobials in generic names, complying with WHO recommendations (16). The hospital uses the Integrated Health Management Information System (IHMIS) for prescribing and has a formulary to guide medicine-related processes, including antibiotics. The practice at BMH is better than primary care and regional hospitals in Tanzania, with DU90% of 84.4% and 87.5%, respectively (12,17). We found the prevalence of AMU to be 26.1%, and items per prescription were 1.4 (recommended 1.6-1.8) (18, 19). The BMH is a referral hospital, so it was expected to have more antimicrobial prescriptions (14). Fortunately, fewer antimicrobial prescriptions were observed among outpatients. The WHO/INRUD drug use indicators recommend the percentage of prescriptions with an antimicrobial to be 20.0-26.8% (18, 19). A recent study in Kenya reported that 30.02% of all prescriptions contained antimicrobials, and an average of 2.9 antimicrobials were prescribed per each prescription encountered (20).
Furthermore, we found that prescriptions including single antimicrobial were 65.3%, two antimicrobials 31.5%, three antimicrobials 2.5%, and four antimicrobials 0.7%. In Pakistan, a study found that 65.1% of prescriptions contained one antimicrobial, 30.6% contained two, and 4.1% contained three. The study suggested that the high prescribing rates of antimicrobials could be due to better clinical outcomes, excessive stock, or aggressive promotion strategies by pharmaceutical companies (21). The comparison was seen in a Jordanian study, which indicated that out of all prescriptions with antimicrobials, 88% had one, 11% had two, and 1% had three antimicrobials (22). The BMH’s antimicrobials prescription rate is nearly two-thirds lower than that of Kenya, Pakistan, and Jordan (20-22).
We observed that antibiotics prescribed at BMH followed the WHO recommendation of no more than two antimicrobials per prescription. This may be attributed to the availability of hospital formulary, clinical auditing, internal and external supervision, and health insurance reimbursement restrictions. These factors greatly influence rational prescribing and contribute to fewer antibiotic prescriptions. This study revealed few prescriptions contained parenteral antimicrobials. The WHO recommends 100% compliance with STG/NEMLIT and 13.4-24.1% of parenteral antimicrobial prescribing (1, 19) In this study, the BMH showed 91% compliance with STG/NEMLIT. The prescribing oral and parenteral antimicrobials was within WHO recommendation. The study in Kiisi county Kenya, displayed 96.7% compliance with national essential medicine list i.e higher than our study. Additionally oral and parenteral antimicrobial prescription was 84.8% and 24.9% respectively (14).
Notably, 96% of prescribed antimicrobials had a clear indication for use, with common indications being post-surgical follow-up, urinary tract infections, and cough. It is concerning that antimicrobials are being used for viral infections and coughs. Fever and cough symptoms are not always due to bacterial infections and often do not require antimicrobial treatment (23). The use of parenteral antibiotics for outpatients is common in tertiary hospitals such as BMH. They are often prescribed for post-surgical follow-up and cancer patients with septic wounds, as well as for those undergoing skin procedures or at risk of acquiring infections. Establishing an Outpatient Parenteral Antimicrobial Therapy Patient Management System (OPAT-PMS) to regulate the use of parenteral antibiotics in outpatients is recommended, as it is not widely implemented in public health facilities in Tanzania. We observed prescriptions of parenteral antimicrobials with a stat and single-dose were 21%, 4% of 24-hour duration, and 3% more than one day. The most common parenteral antimicrobials were ceftriaxone (24%) and metronidazole (19.2%). Other top prescribed antimicrobials included oral metronidazole (8.2%), ciprofloxacin (6.9%), ampicillin/cloxacillin (6.7%), amoxicillin/clavulanic acid (6.5%), flucloxacillin/amoxicillin (6.5%), and azithromycin (5.4%). In Ethiopia, the most commonly prescribed antimicrobials were penicillin G (28.4%), ceftriaxone (24.9%), and cloxacillin (12.84%) (21).
In Tanzania, ceftriaxone is not the preferred first-line treatment for infectious diseases like urinary tract infections due to local evidence of resistance. It has been reclassified as a Watch category antimicrobial. It should generally only be used as a first-line treatment in cases where nitrofurantoin has failed against confirmed severe infections, and sensitivity testing is essential before prescribing (24). The use of ceftriaxone is influenced by its high potency, broad spectrum of activity, low risk of toxicity, and treatment of various bacterial infections. It offers comprehensive pathogen coverage, easy administration, and cost-effectiveness compared to other parenteral antibiotics (23). A similar study found that ceftriaxone, piperacillin/tazobactam, amikacin, and amoxicillin/clavulanic acid were commonly prescribed for pediatric conditions. For post-surgical follow-up in general surgery, obstetrics-gynecology, and orthopedics, cefotaxime, gentamicin, amikacin, and metronidazole were widely prescribed. Tetracyclines are recommended for community-acquired infections (25).
At BMH, four commonly prescribed antimicrobial combinations are ampicillin/cloxacillin, 6.7%, flucloxacillin/amoxicillin 6.5%, ampicillin/cloxacillin 1.6%, and sulfamethoxazole/trimethoprim 0.9%. The preference for these combinations is based on synergism, empirical therapy for poly-microbial infections, and preventing AMR (21, 26). Usually, synergistic effects of antimicrobial combinations are desired when there are high risks of therapeutic failure with individual antimicrobials or a greater probability of developing resistant strains (27). Recent studies refute this argument by reporting synergistic combinations that may enhance the development of resistant strains (4, 28). Other associated risks with antimicrobial combinations include the development of superinfections, more significant toxicity, and an increased financial burden (21). These combinations have also been discouraged by WHO (29).
Our study had some limitations. First, it was conducted over three consecutive months, giving a partial picture of antibiotic use and AMU throughout the year, including seasonal variations. Second, the results cannot be compared to similar local studies in tertiary zonal referral hospitals. Therefore, further prospective or retrospective studies are needed in specialized hospitals in Tanzania to consider seasonal variations.
In conclusion, our study emphasizes the need to strengthen advanced antimicrobial stewardship approaches and recommends further studies in specialized hospitals in Tanzania. Our study further highlights optimal prescribing practices at BMH and recommends instigating the OPAT-PMS at public health facilities to regulate parenteral AMU in outpatients.