Epidemiology
After the World Health Organization (WHO) declared an end to COVID-19 as a global public health emergency on May 5, 2023, we have entered a new era in the pandemic, and most countries have already returned to normal life. This declaration was resulted from the 12-month downward trend of the pandemic, increased immunity due to the highly effective vaccines, decreased death rates, and decreased pressure on once overwhelmed health systems. However, it does not mean that COVID-19 is over as a global health threat, and the pandemic still continues. Indeed, the disease still occurs in very high numbers and causes a significant number of deaths. For example, according to the WHO final report, between July 31 and August 27, 2023, 1.4 million new COVID-19 patients were identified in the world, 55,728 people had to be hospitalized, with an increase of 38% and 40% respectively from the previous month, and 1800 people died due to the disease (1, 2). Current findings suggest that another wave of COVID-19, probably driven by EG.5, has just started all over the world.
Considering that almost all countries in the world do not carry out adequate testing and surveillance, the figures are predicted to be much higher. In China, which was not very affected in the previous waves of COVID-19 and did not have sufficient and effective vaccination, many cases have begun to be seen as of April 2023; that surge was caused mainly by Omicron, XBB.1.5 and as many as 65 million people could become infected per week by the end of June. There are concerns about the emergence of more transmissible and virulent SARS-CoV-2 variants in environments where the disease spreads uncontrollably (3).
In the 4th year of the pandemic, it was seen that COVID-19 caused a new wave with every dominant variant since 2020; two to three waves occurred every single year until now, and the virus still does not show seasonal characteristics like influenza. As a result, as WHO stated on June 13, 2023, COVID-19 is still not seen with a predictable frequency, is still at least two times more deadly than the flu, continues as an epidemic, and has not reached the endemic level yet (4, 5).
Current Circulating Variants of SARS-CoV-2
The Omicron variant of SARS-CoV-2, which has caused at least three different waves with multiple subtypes since its first appearance in December 2021, is still in circulation globally. Currently, the Omicron strains EG.5 and XBB.1.16 are dominant in the world and account for 26.1% and 22.7% of all sequences, respectively, while XBB.1.5, which currently accounts for 10.2% of sequences, continues to decline. EG.5 (publicly known as eris), a descendent lineage of XBB.1.9.2, continues to increase globally, going from 21.1% in week 30 to 26.1 % in week 32. In the USA, in addition to EG.5, the FL.1.5.1 variant is also increasing (6).
Currently, two emerging SARS-CoV-2 variants, called BA.2.86 and FLip variants, including FL.1.5 require close monitoring due to their highly immune-evasive properties. BA.2.86 (publicly known as pirola), classified recently as a “variant under monitoring” because of more than 30 new spike gene mutations compared with the more recently circulating XBB subvariants (XBB.1.5, EG.5.1, FL.1.5.1) (1, 7). BA.2.86 likely originated from an immunocompromised patient who had uncontrolled evolution of the virus. That is another reason why uncontrolled viral growth in immunocompromised hosts should be taken seriously and prevented with efficient antivirals. The large number of mutations and differences from the latest variants in BA2.86 is like the differences between the first Omicron variant (BA.1) and Delta variant (B.1.617.2). It raises concerns about greater escape from existing immunity resulted from vaccines and previous infections compared with other recent variants and the fact that it might lead to extreme number of infections and death like in the first Omicron variant wave of COVID-19 took place at the beginning of 2022. Nevertheless, after the first description on July 24, 2023, BA.2.86 variant has been reported only in a total of 107 samples from 18 countries and has not caused any surge in the countries that it was described except a recently reported limited community transmission in England. As a result, at the moment, BA.2.86 variant appears unable to compete with the other variants, but it is too early to make a strong prediction and this variant should be closely monitored due to its high immune evasion capacities (8-11).
XBB sublineages with F456L followed by L455F mutations called FLip variants. These L and F mutations were predicted in advance as a likely consequence of the widespread SARS-CoV-2 monoclonal antibody treatment for COVID-19 (7). One of the FLip variants, FL.1.5.1 accounts for 13.7% of the COVID-19 cases in the USA at the moment (6).
There is a limited number of studies analyzing the immune escape, transmission, and virulence features of BA2.86 and FLip variants by now, results of those studies suggest that both BA.2.86 and FLip variants (XBB.1.5 + L455F and F456L) are highly immune evasive but FLip variants could be more immune evasive than BA.2.86, patients with an XBB breakthrough infections or reinfections could have higher protection against both of the variants; cell infectivity, therefore infectious capacity of BA.2.86 is lower compared to XBB.1.5 and EG.5; and distinct from XBB variants, monoclonal antibody (mAb) S309 was unable to neutralize BA.2.86, but SA55 remains effective against it (10-13). Mutation profiles of BA.2.82 and FLip variants are not anticipated to impact the performance of current SARS-CoV-2 molecular or antigen-based tests or anti-virals against SARS-CoV-2 (13).
Testing for COVID-19
For diagnosis of acute COVID-19, SARS-CoV-2 testing could be done with an upper respiratory tract nasopharyngeal sample either by using a nucleic acid amplification test (NAAT) or an antigen test.
NAAT-Based Tests
NAAT-based tests are highly sensitive (>95%) and specific (>95%) in diagnosing SARS-CoV-2 infections. The most common type is RT-PCR tests, the gold standard for diagnosing COVID-19. While a positive NAAT result confirms the diagnosis, a negative result generally excludes it; repeat NAAT testing is recommended after 24-48 hours only in the case of high suspicion of COVID-19. NAAT-based tests are recommended first if they are readily accessible and could result in a short period of time. There are both laboratory-based and point-of-care tests. Due to spike mutations that result in S gene target failure, some Omicron subtypes could not be detected in some NAATs rarely, but there is more than one gene target in most of the NAATs; as a result, they still could detect Omicron subtypes. (14-16)
Antigen Tests
Those tests are also recommended for the diagnosis of COVID-19, especially early in the course of symptomatic SARS-CoV-2 infection, because of rapid turnaround time and high specificity. Overall specificity of antigen tests is as high as 99%; as a result, a positive result strongly supports the diagnosis of SARS-COV-2 infection. However, the sensitivity of antigen tests is lower than NAAT-based tests and closely related to the time of infection; sensitivity ranges widely from 81% among patients who are in the first week of symptom onset to 55% among patients who are in the second week of symptom onset and 54% among asymptomatic patients. Repeat testing after 48 hours is recommended to improve sensitivity in the case of the first negative results of cases with symptoms or with recent exposure. Antigen tests generally target the nucleoprotein, so they are expected to detect Omicron subvariants with spike mutations that result in S gene target failure (14-16).
Antigen tests could also be useful for repeated screening of individuals living in crowded, high-risk settings such as nursing homes, schools, etc., aiming to identify patients with SARS-CoV-2 infection quickly and isolate them. This approach is reported to be useful in outbreak settings and if the testing frequency is high enough (17).
The efficacy of antigen testing prior to events and gatherings has not been proven, especially if additional preventive measures were taken along with testing. To define the time for discontinuation of infection prevention precautions among patients with SARS-CoV-2 infection, time and symptom-based criteria are recommended for immunocompetent patients with asymptomatic or mild to moderate disease. Nevertheless, a test-based strategy is more suitable among patients who are either immunocompromised or have a severe/critical illness. A NAAT or antigen test can be used for a test-based strategy. For persistently positive patients, either cycle threshold values of NAATs or determination of antibody level against SARS-CoV-2 have been used to assess infectivity and guide clinical management, but those strategies are not validated and proved to be effective (14-16).
Currently, testing for SARS-CoV-2 on nasopharyngeal swab recommended for those;
- with appropriate symptoms and/or signs of COVID-19 (fever, cough, shortness of breath, muscle aches, sore throat, and/or loss of taste or smell)
- with a history of contact with a patient with COVID-19 in the last two weeks
Routine preoperative SARS-CoV-2 testing is not recommended in patients without contact and symptoms. Instead, perioperative screening for symptoms and contact with COVID-19, robust infection control measures, and targeted testing are recommended according to the community incidence of COVID-19 and the infrastructure facilities of the institution.
SARS-CoV-2 PCR testing can be considered in the following groups according to facilities of the institutions and in hospitals that cannot fully implement the specified key measures given in the “COVID-19 Prevention at the Hospitals” section:
- Immunocompromised patients admitted to the hospital or patients who will be admitted to wards for immunocompromised patients.
- Patients undergoing major surgery (e.g., cranial, cardiac, thoracic, abdominal surgical interventions, implantation of prosthesis, or any procedure associated with extensive tissue dissection or transection).
Prevention of COVID-19
Prevention of COVID-19 relies on vaccination, hand and respiratory hygiene, surveillance, avoiding enclosed spaces with poor ventilation, proper indoor ventilation, isolation of COVID-19 patients, avoiding close contacts with patients who had respiratory symptoms and mask usage. Close contacts of COVID-19 patients should be monitored for symptoms, wear a mask, and get tested for SARS-CoV-2 in the presence of symptoms.
Preventive Measures
COVID-19 Prevention in the Community Settings
In addition to well-known key interventions, including isolation, vaccination, and proper ventilation, the following recommendations could be taken into consideration (18, 19):
1. Mask Usage: Recommendations for using masks in public places depends on local epidemiological trends and hospitalization levels of COVID-19, percent of patients fully vaccinated against the disease, total level of immunity in the community, and the setting in which people find themselves.
At the current level of the Pandemic, mask usage is recommended;
- For those who have cold or flu-like symptoms (such as malaise/fatigue, fever, runny nose, sore throat, headache, muscle aches, cough, shortness of breath, sore throat, and/or loss of taste or smell) or who test positive for SARS-CoV-2.
- When being in a place with a person having COVID-19 or like-illness.
- In enclosed, crowded, or poorly ventilated places.
- Following a recent exposure to COVID-19.
- For patients at high risk of severe complications from COVID-19 (people aged ≥60 years; or those with underlying serious comorbidities, such as cardiovascular disease or diabetes mellitus, chronic lung disease, cancer, cerebrovascular disease, immunosuppression, obesity, or asthma; unvaccinated and pregnant).
At the current level of the Pandemic, mask usage could be beneficial;
- If COVID-19 incidence in the community is high or rising.
- If not vaccinated for COVID-19 or COVID-19 vaccination coverage is low at the community level.
- If population immunity level to SARS-CoV-2 is low.
- In the case of greater crowding and poorer indoor ventilation.
2. Maintaining and strengthening COVID-19 surveillance, including genomic surveillance, is of utmost importance.
COVID-19 Prevention at the Hospitals
In addition to well-known key interventions including isolation, vaccination, and proper ventilation, those are the current recommendations for mask usage in healthcare facilities (18, 19):
- In the case of community or cluster SARS-CoV-2 transmission: Universal masking for everyone, including staff, visitors, outpatients, etc., is recommended both in clinical and common areas, including cafeteria and staff rooms.
- In the case of sporadic SARS-CoV-2 transmission: Targeted continuous medical mask use in clinical areas (when caring for non-COVID-19 patients and during routine activities apart from when eating and drinking) but not in non-patient areas is recommended.
Additional Recommendations for Surgical Patients
Unvaccinated patients who will undergo surgery should be properly vaccinated, with the last dose ending two weeks before surgery.
Due to high risk of morbidity and mortality among patients having COVID-19 during or just before the major surgical procedures, it is still recommended that elective surgical interventions be postponed for seven weeks after an acute COVID-19 infection, if possible. Additionally SARS-CoV-2 testing is still recommended for patients scheduled for elective major surgery (20).
COVID-19 Vaccines
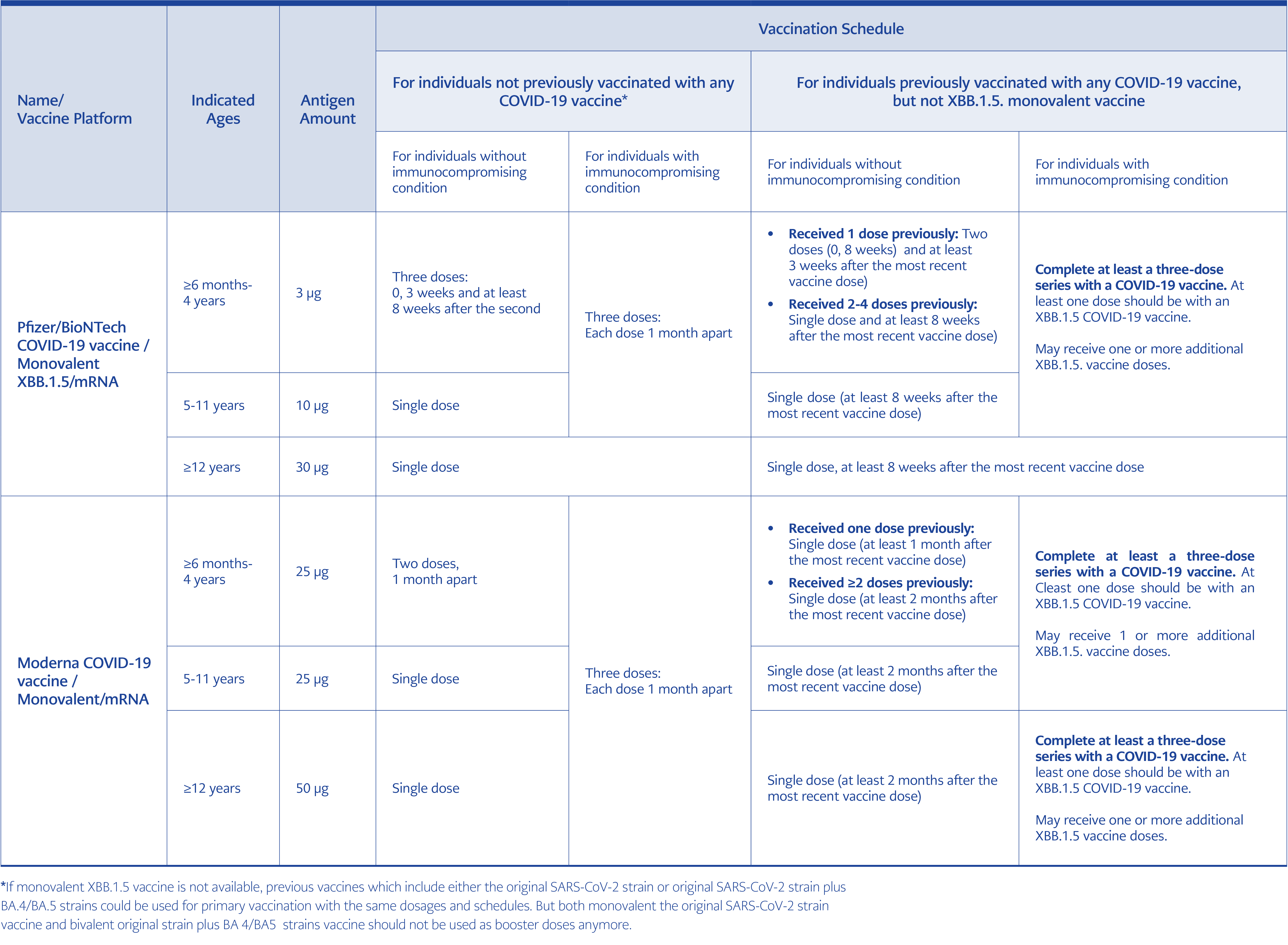
The first introduced vaccines to prevent COVID-19 were monovalent and included the original SARS-CoV-2 strain. Those first version vaccines, of which approximately 14 billion doses have been administered so far, have been particularly effective (over 90%) in preventing severe illness and death and have saved the lives of more than 20 million people so far (21). In the latest report of the UK Health Security Agency (UKHSA), which regularly monitors the efficacy and safety of COVID-19 vaccines, it was reported that the benefits of COVID-19 vaccines far outweigh the potential risks (22). However, vaccine protection against SARS-CoV-2 infection decreased over time because of new emerging immune-evasive variants. Protection against hospitalization and death also decreased. In order to increase the effectiveness of those vaccines, bivalent mRNA vaccines, including original strain plus BA.4-BA.5 Omicron variants, were developed in June 2022. Then, in high-income countries such as the USA, one, two or more booster doses of those bivalent vaccines have been administered in defined risk groups starting from October 2022 (22-24). The Technical Advisory Group on COVID-19 Vaccine Composition (TAG-CO-VAC), which evaluates many developments such as the course of the disease, its frequency, distribution, the status of existing variants, and the effect of old vaccines on existing variant infections, stated that the content of the new COVID-19 vaccine should be monovalent and included strains derived from XBB.1 such as XBB.1.5 or XBB.1.16. This recommendation was based on the fact that antibodies formed with existing bivalent vaccines could not sufficiently neutralize the XBB.1-derived strains and that repetitive applications of the original strain could be associated with a decreased immune response (namely immune imprinting) against new antigens (25). XBB.1.5 strain containing new monovalent COVID-19 vaccines have already been produced and recently been approved by the US Food and Drug Administration (FDA) for the vaccination of all people aged six months and older against COVID-19 during the autumn of 2023 (25-27).
Studies have shown that bivalent vaccine boosters are more effective than the original monovalent vaccines for preventing death and hospitalizations due to COVID-19. In the latest data from the U.S. Centers for Disease Control and Prevention (CDC), mortality rates were 16.2, 11.5, and 5.3 times higher in the Delta, BA.1, and BA.2/ BA.4/BA.5 periods, respectively in the unvaccinated compared to vaccinated persons. The mortality rate in the unvaccinated was 14.1 times higher than in those vaccinated with the bivalent vaccine, and mortality was 2.6 times higher in those vaccinated with the original monovalent vaccine in the BA.4/BA.5 period than in those vaccinated with the bivalent vaccine. The mortality rate in the unvaccinated persons was 23.7 times higher in the 65-79 age group and 10.3 times higher in the ≥80-year age group compared to the bivalent supplementary dose group; when compared with the original monovalent supplementary dose group, it was 8.3 times higher at 65-79 years and 4.2 times higher at ≥80 years old groups. These data showed that the death protection rate of bivalent vaccines was higher than that of original monovalent vaccines in the Omicron period. According to the data of April 1, 2023, in the USA, although the protection rate of both bivalent and monovalent vaccines against death has decreased, it still continues (28, 29).
In the UK’s latest vaccine efficacy report, the efficacy of vaccines against death in persons >65 years of age was calculated as 56.9% at week >40 for those given a reminder dose and 68.2% at week >20 for those given two reminder doses. Bivalent vaccines administered in autumn 2022 have been shown to provide an additional 47% efficacy against hospitalization at age >50, 10 weeks post-vaccination. In the same report, it was concluded that COVID-19 vaccination with both primary and booster doses is the safest and most effective way to protect pregnant women from severe COVID-19. It was observed that 70-75% of pregnant women who gave birth in England were vaccinated against COVID-19 in at least two doses, and 48% were vaccinated for three doses (22).
Monovalent XBB.1.5 containing vaccine was found to elicit potent neutralizing antibody response against Omicron XBB related variants including XBB.1.5, XBB.1.6, XBB.2.3.2 as well as more recent EG.5.1, FL.1.5.1 (31). Additionally, laboratory data suggest the new updated vaccines could generate enough neutralizing antibodies against BA.2.86 variant (10-12, 27).
In parallel with the increase in the number of people who have been vaccinated and who have had the disease, the WHO Strategic Advisory Group of Experts on Immunization (SAGE) has updated its COVID-19 vaccination recommendations. SAGE has identified the elderly, the young with comorbidities (such as diabetes and heart disease), the immunocompromised, including children >6 months old (such as individuals living with HIV and transplant recipients), pregnant women, and healthcare workers as high-priority groups for COVID-19 vaccination. For those groups, an additional booster dose 6-12 months after the last dose of primary vaccination is recommended. Those aged <50-60 years without comorbidities were considered an intermediate-risk group for COVID-19 and only a single dose booster is recommended to them after primary vaccination. Young people aged <18 years and adults without comorbidity are grouped as low risk, leaving the decision to vaccinate against COVID-19 for these individuals to the countries themselves (30). Europe and the USA recommend primary COVID-19 vaccination for all children aged >6 months and adults.
High-income countries such as the USA and UK abandoned the use of original strain monovalent vaccines and original strain plus BA4/BA5 bivalent vaccines due to the fact that the effectiveness of those vaccines against existing variants is lower, and “immune imprinting” could be a problem. At the moment, XBB.1.5 containing monovalent SARS-CoV-2 vaccines are recommended for both revaccinations and primary vaccinations, and the former vaccines are no longer recommended in those countries (21, 27). However, XBB.1.5 containing vaccines will not be available in most lower and middle-income countries by this autumn. Due to the fact that COVID-19 vaccines based on the original virus continue to provide some protection against severe disease, those vaccines could be used especially for primary vaccinations of high-risk groups in settings where updated vaccines are not available (30) (Table 1).
Elimination of SARS-CoV-2 from the community is not expected due to the disappearance of COVID-19 isolation measures and the decrease in herd immunity against mild disease 1-2 weeks after the last dose of vaccine. The main purpose of vaccination is to reduce deaths and hospitalizations from COVID-19. As a result, most countries like China, France, the UK, Germany, and Mexico recommend COVID-19 boosters for only individuals at higher risk for severe COVID-19 (27). Due to the nearly 40% decrease in long-COVID conditions among vaccinated individuals (32), some authors and countries like the USA recommend booster vaccination for everyone older than six months to decrease the risk of transmission, the occurrence of not only COVID-19, but also long COVID-19 conditions (25, 27, 32).
Developed and updated vaccines for COVID-19 are still being used due to their success in protecting against severe disease, but research continues to make a pan-coronavirus vaccine that will not be affected by variants.
Monoclonal Antibodies
Pre-exposure Prophylaxis
A long-acting monoclonal antibodies (mAbs) combination tixagevimab/cilgavimab has been used effectively for pre-exposure prophylaxis against COVID-19 in immunocompromised patients or in patients who are unable to receive vaccination. Although some Omicron variants, including BA.1 and BA.2, could be neutralized to some extent by tixagevimab/cilgavimab, it is not active against current circulating Omicron subvariants including XBB.1.5, XBB.1.16 and EG.5, for this reason it is no longer recommended for pre-exposure prophylaxis of COVID-19 (33, 34).
Cardiac serious adverse effects were more frequently seen in tixagevimab/cigavimab group than in the placebo in a post-hoc analysis of data from the phase-3 trial, and the FDA recommended including a warning about those findings (35). Nevertheless, in a recent retrospective propensity-matched cohort study, no increased risk of cardiovascular events was seen up to 90 days after tixagevimab/cilgavimab administration, including in patients with pre-existing cardiovascular disease (36).
Postexposure Prophylaxis
In a randomized controlled trial (RCT),when using as postexposure prophylactic treatment, tixagevimab/cilgavimab did not show a significant effect on the RT-PCR-positive symptomatic COVID-19, with a reduction rate of 33.3% (95% confidence interval [CI]=-25.9-64.7; p=0.21), but in the same study it was shown to reduce RT-PCR-positive symptomatic COVID-19 by 73.2% who were SARS-CoV-2 RT-PCR-negative (n=974, 87%) or missing at baseline. Unfortunately, tixagevimab/cilgavimab is not active against circulating Omicron subvariants and has never been authorized or recommended for post-exposure prophylaxis of COVID-19 (37).
Casirivimab-imdevimab or bamlanivimab-etesevimab were previously approved for the post-exposure prevention of SARS-CoV-2 infection in unvaccinated and immunocompromised patients. However, these combinations can not neutralize the Omicron subvariants and not recommended anymore for that purpose.
BCG for Prevention of COVID-19
As bacillus Calmette–Guérin (BCG) vaccine was shown to have some immunomodulatory effects due to training immunity and to be associated with a reduced risk of respiratory infections previously, it was proposed that BCG vaccination could be used to protect from COVID-19 early in the pandemic while no specific vaccine was available. However, two placebo-controlled RCTs, including a total of 3312 healthcare workers, could not show the efficacy of BCG for the prevention of COVID-19 (38-41).
But, in a recent placebo-controlled RCT study of 2064 healthy elderlies analyzing the effect of a genetically modified BCG vaccine, VPM1002, on the mean number of days with severe respiratory infections including COVID-19 at hospital and/or at home in a total of 240 days, VMP1002 was found to be associated with a significantly lower rate of self-reported fever (5.3% vs. 2.5%, odds ration [OR]=0.46, 95% CI=0.28-0.74); p=0.001), but not a significant rate difference of severe respiratory disease at hospital and/or at home (3.8% vs. 3.1%, OR=0.81, 95% CI=0.50-1.32); documented SARS-CoV-2 infection was lower among VPM1002 recipients, but difference was not significant (0.8% vs 0.2%; p=0.05) (42). Additional studies are needed to define the role of BCG and its derivatives in the prevention of respiratory tract infections, including COVID-19.
Treatment of COVID-19
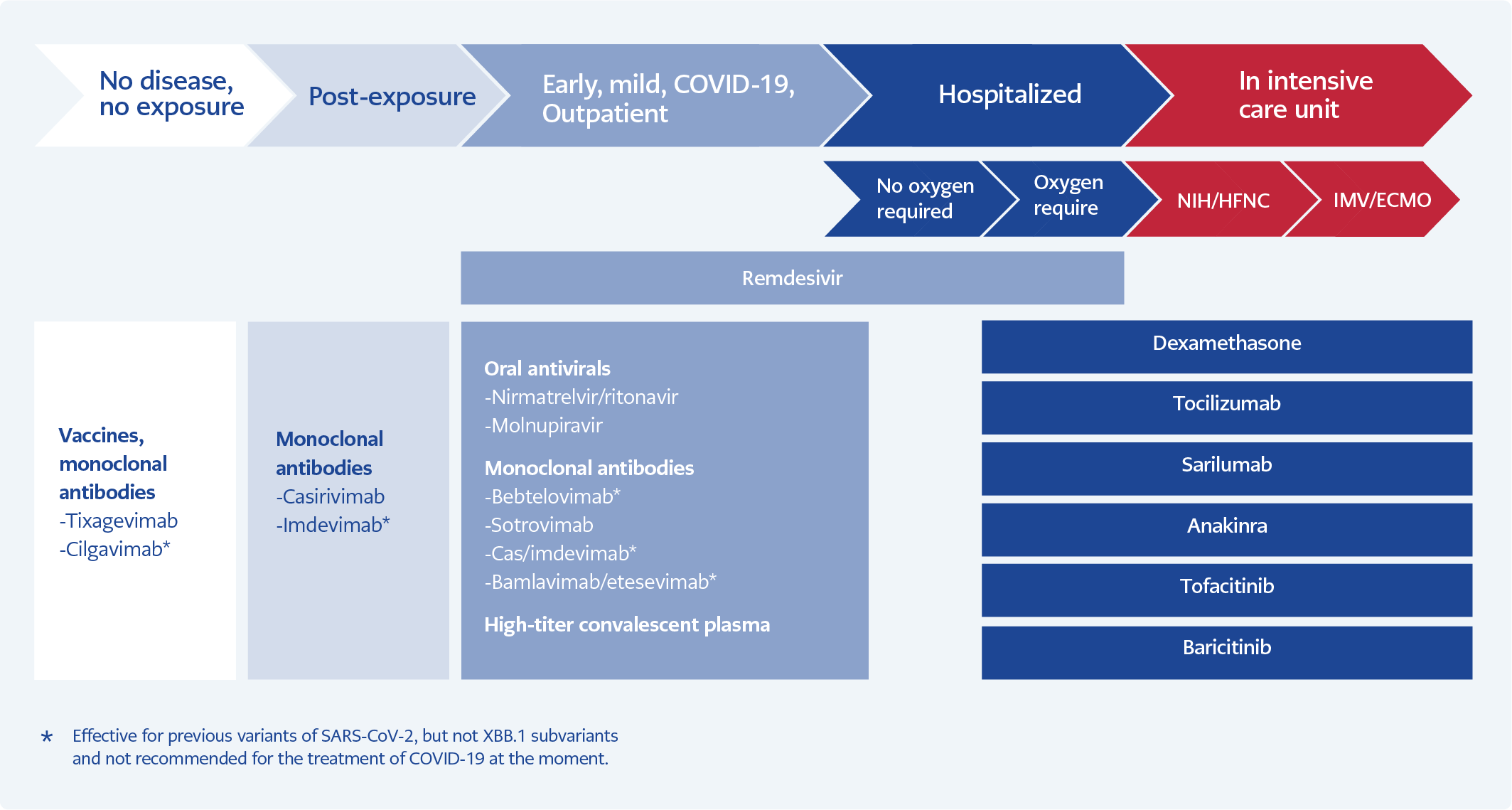
Immunity acquired by either vaccinations or getting the disease have significantly decreased the mortality rates in COVID-19, from 0.6-1% to 0.06-0.1% in all groups and from 15.1% (Delta) to 4.9% in hospitalized patients. Currently, those who die from COVID-19 appear to be older patients (>65 years), having ≥3 comorbidities or immunocompromised. Hospitalization rates also dropped significantly. Efficacy studies of the antivirals currently used to treat COVID-19 have been conducted based on outcomes of death and/or hospitalization; however, because of current lower rates of hospitalization and/or death from the disease, studying drugs according to these outcomes is not likely. It is necessary to identify new outcomes to analyze the effectiveness of the drugs against COVID-19, such as time to recovery or viral clearance (43). For the treatment of COVID-19, different drugs acting on different pathological conditions are required according to the stage of the disease. While antiviral agents are needed mainly during the early phase of the disease and generally recommended for patients with mild illness and symptoms in the first 5-7 days, anti-inflammatory drugs, including dexamethasone and anti-cytokines, are recommended for hospitalized patients with more severe disease (Figure 1).
Anti-Viral Agents
Convalescent Plasma
In an individual participant data meta-analysis of 5 RCT, among outpatients with COVID-19, treatment with convalescent plasma (CP) reduced the rate of all-cause hospitalization from 12.2% to 8.5% with a 30.1% relative risk reduction. The hospitalization reduction was greatest for those having either early transfusion or high titer plasma (44). In another systematic review and meta-analysis of three RCTs enrolling 1487 participants and five controlled studies compared with the controls, CP transfusion was also shown to be associated with a decrease in mortality with a relative risk ratio (RR) of 0.63 (95% CI=0.50-0.79) for patients who are immunocompromised and have COVID-19 (45). Infectious Diseases Society of America (IDSA) recommends a high-titer COVID-19 convalescent plasma within eight days of symptom onset for ambulatory COVID-19 patients with mild-to-moderate COVID-19 at high risk for progression to severe disease who have no other treatment options (nirmatrelvir/ritonavir, remdesivir, molnupiravir and SARS-CoV-2 specific mAbs) (46).
In the meta-analysis of 19 RCTs including 17,021 patients, no survival benefit of CP was seen in severely or critically ill, hospitalized immunocompetent COVID-19 patients; as a result, CP is not recommended for the treatment of patients hospitalized for severe COVID-19 at the moment (47). Nevertheless, for the treatment of immunocompromised patients hospitalized for COVID-19, limited data suggest that high-titer convalescent plasma may have a role, especially if the patient had deficits in antibody production (45, 47).
Monoclonal Antibodies for SARS-CoV-2
Bamlanivimab, casirivimab-imdevimab, sotrovimab, and bebtelovimab were effective options for treating high-risk COVID-19 outpatients during the previous waves of COVID-19. Among high-risk patients, nearly 8000 outpatients with a positive SARS-CoV-2 test result, the composite outcome of hospitalization or death at 28 days was lower in 2571 patients treated with mAbs (4.6%) than patients not treated (7.6%) (RR=0.61, 95% CI=0.50-0.74); the RRs were 0.55, 0.53, and 0.71 when the Alpha, Delta, and Omicron variants were presumed to be predominant, respectively and among immunocompromised patients, the RR was 0.45 (95% CI=0.28-0.71). But the rapid emergence of new variants of SARS-CoV-2 requires continuous evaluation of mAbs preparations (48, 49).
Unfortunately, current circulating XBB.1 Omicron subvariants, including XBB1.5, XBB 1.16 and EG.5, escape all clinically available anti-SARS-CoV-2 mAbs, and there is not an available mAb for the treatment of COVID-19 at the moment. Only C1717, S3H3, S309 (sotrovimab), and 10-40 showed relatively little fold change in neutralizing activity against XBB1.5 and (49); for this reason, the UK National Health Service (NHS) recommends sotrovimab only for high-risk COVID-19 outpatients for whom nirmatrelvir/ritonavir is contraindicated or unsuitable (50).
Nirmatrelvir/Ritonavir
After finding that it reduced hospital admission and mortality by 89% for unvaccinated high-risk patients with mild COVID-19 at the first five days of their symptoms, nirmatrelvir/ritonavir was recommended as the first-line antiviral for mild COVID-19, but not for severe or hospitalized cases (46). Later on, several studies showed that nirmatrelvir/ritonavir was also effective in reducing mortality and hospital admission among vaccinated high-risk patients infected with the Omicron variant by 85% and 55%, respectively (51, 52). Nevertheless, in a recent open-label RCT study of 164 mild-moderate, high-risk, and mostly unvaccinated -COVID-19 patients from China, nirmatrelvir/ritonavir was not found to be more effective than standard care in decreasing mortality or increasing viral clearance by day 7 and 14 during Omicron BA2.2 circulation (53). This result could be related to including patients with more advanced disease.
Nirmatrelvir/ritonavir was found to be well-tolerated and caused no adverse effect for both mother and baby in a study of 47 pregnant women with COVID-19, mostly with additional comorbidities. Considering the higher risk of morbidity and mortality from COVID-19 in pregnant women, nirmatrelvir/ritonavir could be used in pregnant women with COVID-19, especially for unvaccinated ones (54).
A modified dose of nirmatrelvir/ritonavir was shown to be effective and well tolerated with a good safety profile among patients with COVID-19 and chronic kidney disease, including advanced stages with dialysis (55).
Treatment with nirmatrelvir/ritonavir during acute COVID-19 was also found to be related to reducing post-acute sequels of the disease and post-acute hospitalization with a hazard ratio (HR)=0.52 (0.35, 0.77) and HR=0.70 (0.61, 0.80), respectively in a retrospective analysis (56). There are concerns about the development of resistant SARS-CoV-2 against nirmatrelvir/ritonavir. In a cell-culture study, mutations conferring high level (E166V and L50F + E166V) resistance and fitness in infectious systems (L50F and L50F + E166V) were developed against SARS-CoV-2 under nirmatrelvir/ritonavir effect (57). In another study, it was shown that SARS-CoV-2 three CL protease mutants selected in a vesicular stomatitis virus-based system, and those mutants are cross-resistant to ensitrelvir and GC376 and most of those resistance mutations already existed in SARS-CoV-2 sequences of Global Initiative on Sharing All Influenza Data (GISAID) and National Center for Biotechnology Information (NCBI) databases (58).
Another problem after nirmatrelvir/ritonavir usage for COVID-19 treatment is viral and symptom rebounds, which have been described several times, with unknown reasons. In a recent prospective cohort study of COVID-19 patients who accepted and did not accept the use of nirmatrelvir/ritonavir, viral and symptom rebounds were found to be 14.2% vs. 9.3%, which were not significantly higher than the control groups (p=0.41), (p=0.06) (59). Large studies are needed to better understand those viral and symptom rebounds after nirmatrelvir/ritonavir usage for COVID-19.
Remdesivir and Oral Analogues of Remdesivir
A three-day course of intravenous (IV) remdesivir was found to be effective in reducing hospital admission and mortality by nearly 90% among COVID-19 outpatients and recommended as a first-line treatment in the guidelines (46). In a recent propensity score matching adjusted control cohort study of patients hospitalized for COVID-19, including 24,856 remdesivir and 24,856 non-remdesivir patients, mortality rates were significantly lower for patients treated with remdesivir, with a 17% reduction (HR=0.83, 95% CI=0.79-0.87) (60). In a systematic meta-analysis of eight RCTs, including 10,854 patients hospitalized for COVID-19, mortality rate on the 28th day was found to be significantly lower in patients treated with remdesivir (12.5%) than in patients not treated with remdesivir (14.1%), with an adjusted OR (aOR)=0.88 (%95 CI=0.78-1.00, p=0.045) (61). As a result, remdesivir is also recommended for the treatment of hospitalized patients with severe COVID-19 but not with critical illness (46).
In phase-3 RCT of VV116, an oral analog of remdesivir, sustained recovery time was found to be noninferior to nirmatrelvir/ritonavir, with fewer safety concerns among high-risk COVID-19 patients (62). As IV treatment could be very difficult in outpatient settings, oral remdesivir analogs would be promising alternatives for the treatment of COVID-19.
Molnupiravir
After finding that it was effective in reducing hospitalization and/or mortality from COVID-19 by 30% among unvaccinated high-risk mild COVID-19 outpatients in the phase 3 RCT, molnupiravir was recommended for the treatment of mild COVID-19 in the case of unavailability of nirmatrelvir/ritonavir or remdesivir (46). The UK PANAROMIC trial (The Platform Adaptive Trial of Novel Antivirals for Early Treatment of COVID-19 in the Community) study of vaccinated outpatients with COVID-19 revealed that, compared to standard care, molnupriavir use did not decrease the rate of hospitalization or death, but associated with faster time to recovery and viral clearance on the seventh day of treatment (63). In a later study, the effect of molnupiravir and nirmatrelvir/ritonavir were analyzed in three retrospective target trial emulation studies, including nearly 250,000 non-hospitalized veterans in the USA during the Omicron wave. Both nirmatrelvir-ritonavir and molnupiravir were found to be effective in reducing 30-day death with an RR=0.23 for both drugs (95% CI=0.13-0.41 and 95% CI=0.13-0.43, respectively) (64). Finally in a recent real-world study including 1,074,856 non-hospitalised patients with COVID-19, molnupiravir use was associated with lower risk of death (HR=0·76, 95% CI=0.61-0.95) and in-hospital disease progression (HR=0·57, 95% CI=0.43-0.76) than non-use, whereas risk of hospitalization was similar in both groups (HR=0.98, 95% CI=0.89-1.06]) (65).
Finally, in a recent nationwide, observational study of 780 patients with COVID-19 and an immunocompromising conditions, using of nirmatrelvir/ritonavir or molnupiravir was associated with a significantly lower rate of the composite outcome of hospitalization, death and intensive care unit (ICU) admission with an OR of 0.37 (95% CI=0.22-0.61). These findings support the use of either oral antiviral in this population (66).
There are some concerns about the mutagenic effect of molnupiravir; it was not recommended for pregnant women and children and was shown to induce single nucleotide polymorphisms (SNPs) randomly during treatment, but these SNPs rarely persist and limit the risk of selection of viral variants (67). Nevertheless, during the treatment of immunocompromised patients, some of those mutations could persist and, in some cases, were fixed in the virus population, which has the potential to generate new variants and prolong the pandemic (68).
Molnupiravir was not approved for the treatment of COVID-19 by the European Union (69) and was not recommended for treating COVID-19 in the UK because of its lower rate of effectiveness and cost (50). Nevertheless, IDSA recommends molnupiravir for patients with mild-to moderate COVID 19 and at high risk for progression to severe disease just in the case of no other treatment options (70).
Ensitrelvir Fumarate
Ensitrelvir fumarate, a SARS-CoV-2 protease inhibitor, resulted in a significantly higher rate of viral clearance rate than placebo, both in patients with COVID-19 treated with both 125 mg and 250 mg doses of the drug. It was also shown to be associated with quicker clinical improvement compared to the placebo (71-73).
Favipiravir
Favipiravir was shown to be ineffective for treating COVID-19 in the previous RCTs performed during early years of pthe pandemic. Although there are some new observational (74) and RCTs (75-77) showing some beneficial effect of favipiravir on clinical recovery or viral clearance in patients with COVID-19, no benefit on survival or on progression to severe disease or hospital admission (78, 79) or clinical recovery and/or viral clearance (80-84) have been shown in most of the recent RCT’s performed in patients with COVID-19 in both hospitalized and outpatients. As a result, favipiravir is not recommended for the treatment of COVID-19 in the current guidelines.
Pegylated Interferon Lambda
Pegylated interferon lambda, with a single 180 μg dose, was found to reduce hospital or emergency unit admission of mostly vaccinated high-risk patients with COVID-19 by an
RR of 0.49 (95% CI=0.30-0.76) compared to placebo (85). In another RCT of 212 high-risk adults hospitalized for COVID-19, clinical recovery was quicker in patients treated with interferon beta-1b and remdesivir combination than patients treated with only remdesivir (4 vs 6.5 days; HR=6.59, 95% CI=6.1-7.09; p<0.0001) when compared to the control group (86). Although more studies are needed to define the place of interferons in the treatment of COVID-19, after the availability of direct-acting anti-viral agents against SARS-CoV-2, it has become very difficult to carry out those studies.
Vitamins and Minerals
A couple of observational studies reported an association between some mineral and vitamin deficiencies and higher risk or severity of COVID-19 (87, 88). However, in a recent RCT, treatment with high-dose zinc, vitamin C, or both was not found to be related with the decreased duration of COVID-19 symptoms compared with standard of care (89). In another placebo-controlled RCT of 470 patients with COVID-19 without organ failure (40.4% were ambulatory and 59.6% were hospitalized), death or ICU admission was lower in the zinc group versus the placebo group (OR=0.58, 95% CI=0.33-0.99) (90). Potential explanations for the conflicting results between those two RCTs can be lower dietary zinc intake in Tunisia than in the USA, the doses of zinc used in the studies, and differences in comorbidities and severity of COVID-19 in the included patients. Additional studies are needed to define the exact role of zinc for the treatment of COVID-19 (91).
In the previous studies, it was shown that oral zinc formulations may shorten the duration of symptoms of the common cold, but there was significant heterogeneity among those studies and high-quality studies are needed before definitive recommendations for clinical practice can be made. As zinc-containing intranasal products can cause irreversible anosmia, they should not be used (91-93).
Vitamin D supplementation also has not been shown to be beneficial for COVID-19 outcomes and there is no strong evidence that vitamin D supplementation reduces the risk or severity of COVID-19 (94, 95).
Metformin, Ivermectin, or Fluvoxamine
Metformin, ivermectin or fluvoxamine were found to be ineffective in reducing hospitalization or death from COVID-19 in several RCTs (96, 97). Nevertheless, in a recent placebo-controlled RCT of 738 patients with early-onset COVID-19, oral fluvoxamine plus inhaled budesonide was associated with a lower risk of emergency unit or hospital admission than the placebo 1.8% (95% credible interval [CrI]=1.1%-3.0%) vs. 3.7% (95% CrI=2.5%-5.3%]; relative risk 0.50 [95% CrI= 0.25 to 0.92) (98). Additional studies are needed.
Anti-inflammatory and Immune-Modulating Agents
Anti-inflammatory and immune response modulating agents, including corticosteroids and anti-cytokine agents, are useful for patients with severe disease, which occurs later in the disease course of patients with COVID-19.
Corticosteroids
Dexamethasone 6 mg/day for patient on oxygen because of severe COVID-19 was shown to be effective in reducing mortality from the very beginning of the Pandemic. If dexamethasone is unavailable, other glucocorticoids with equivalent doses can be used.
Higher Doses of Corticosteroids
In the Randomised Evaluation of COVID-19 Therapy Trial (RECOVERY Trial), dexamethasone 20 mg/daily significantly increased the risk of death compared with usual 6 mg daily dexamethasone care in patients hospitalized for COVID-19 with clinical hypoxia who required either no oxygen or simple oxygen only (99). Daily 80 mg prednisolone was also found to be not more effective for reducing mortality from COVID-19 than daily 6 mg dexamethasone in another RCT including 677 patients with COVID-19 pneumonia requiring oxygen (nearly %50) or noninvasive respiratory support (remaining 50%) (100).
In a recent RCT enrolled 1000 patients with COVID-19 receiving either at least 10 L/min of oxygen or noninvasive ventilation, or invasive ventilation, 12 mg daily dexamethasone resulted in lower 28-day mortality (27% versus 32%, adjusted risk ratio [aRR]=0.86, 95% CI=0.68-1.08) compared with 6 mg daily, but the difference was not statistically significant (101). Other smaller studies have not found a reduction in mechanical ventilation (MV) or mortality rates with high- versus lower-dose dexamethasone (102-105). Current data suggest that higher doses of dexamethasone could be harmful for COVID-19 patients receiving low-flow oxygen; higher doses could be more beneficial than the standard dose of dexamethasone for COVID-19 patients on higher-flow oxygen or MV. The threshold oxygen flow rate for higher doses of dexamethasone is yet to be determined, but it could probably be greater than 10 L/min, which was the dose used in one of the biggest studies investigating the effect of a higher dose of dexamethasone (12 mg/daily) for severe COVID-19. There will be more evidence about the place of higher dexamethasone doses in the treatment of severe COVID-19 after the availability of results of the subgroup analysis of patients on ventilator or extracorporeal membrane oxygenation (ECMO) in the RECOVERY RCT (99-102).
Inhaled Corticosteroids
Some evidence suggests that inhaled corticosteroids, including budesonide or ciclesonide, may affect the rate of hospital admission and death among patients with mild COVID-19. In a meta-analysis of three RCTs including 3607 patients of whom 2490 had mild infections, inhaled corticosteroids (budesonide 860 patients; ciclesonide 197 patients) were found to reduce admission to hospital or death (at up to 30 days) (RR=0.72, 95% CI=0.51-0.99); and to increase resolution of symptoms at day 14 (RR=1.19, 95% CI=1.09-1.30) (106).
In high-risk patients with early/mild COVID-9 who are able to use inhaler devices, inhaled corticosteroids could be used to decrease more severe outcomes, including death and hospitalization, despite the increased risk of the resolution of symptoms.
Anti-Cytokine Agents
Interleukin-6 Pathway Inhibitors: Tocilizumab, Sarilumab
In the meta-analysis of 18 RCTs including more than 7000 patients with severe COVID-19 the majority of whom were already on corticosteroids, compared to placebo/standard care, IL-6 inhibitors were shown to be associated with decreased all-cause mortality rate at day 28 with an average OR of 0.78 (95% CrI=0.65-0.94) and relative risk of 0.88, (95% CI=0.81-0.94) for tocilizumab; and 0.91 (95% CrI=0.60-1.40) for sarilumab. Interleukin (IL)-6 receptor antagonists also had a greater than 99.9% probability of improving 6-month survival (adjusted HR [aHR]= 0.74, 95% CrI=0.61-0.90) (107, 108). Besides rreducing mortality during the acute phase of infection, tocilizumab also improved survival six months after acute COVID-19 (109).
Interleukin-1 Inhibitors: Anakinra, Canakinumab
Interleukin (IL)-1 inhibitor anakinra received an emergency use authorization from the FDA for use in patients hospitalized with COVID-19 and who require oxygen support and who are at higher risk of progression to severe disease and have an elevated plasma soluble urokinase plasminogen activator receptor (suPAR). This approval followed the positive results of an RCT of anakinra in patients with severe COVID-19, in which serum levels of the suPAR were used as a guide for the initiation of treatment with anakinra (110).
A recent meta-analysis of 23 studies (five RCTs, 30 cohort studies, eight case series, and one preliminary study) including 3179 COVID-19 patients showed a statistically significant difference in duration of ICU stays [MD= -0.65, 95% CI=-1.09 – -0.03); p=0.04], the number of patients who needed ventilator support (RR=0.57, 95% CI=0.39-0.84); p=0.004), and mortality rate (RR=0.80, 95% CI=0.66-0.99; p=0.04) (111) among patients treated with anakinra. Despite those positive results, several RCTs and meta-analyses of RCTs did not find any survival benefit by using either anakinra or canakinumab, another IL-1 antagonist, in the treatment of COVID-19 (112-114).
The positive effect of anakinra on the clinical outcome of patients with COVID-19 seems to be related to the timely administration of it. As suPAR is not widely available in most countries at the moment, some easy-to-perform tests such as CRP, ferritin, and neutrophil-to-lymphocyte ratio ≥7 should be investigated in additional clinical trials as a surrogate of elevated suPAR.
Janus Kinase Inhibitors: Baricitinib, Tofacitinib, Ruxolitinib
In the meta-analysis of nine RCTs including 11,888 patients with severe COVID-19 and 1485 deaths, using of Janus kinase (JAK) inhibitors, including baricitinib or tofacitinib, were associated with a significant reduction in mortality with a rate ratio of 0.80 (0.72-0.89; p<0.0001) (115).
In another systematic review and meta-analysis of four RCTs including 10,815 patients hospitalized for severe COVID-19, baricitinib treatment was found to be associated a statistically significant reduction in 28-day mortality (OR=0.69, 95% CI=0.50-0.94; p=0.04, I2 =65%) and composite outcome of progression to severe disease needing positive pressure ventilation, invasive mechanical ventilation (IMV) or death (OR=0.89, 95% CI=0.80-0.99, p=0.03, I2=0%) (116). Baricitinib was found to be non-inferior to tocilizumab regarding the composite outcome of death or IMV by day 28 among 251 patients with severe COVID-19 patients included in an RCT (117).
Tofacitinib, another JAK inhibitör, was also shown to be effective for reducing composite outcome of respiratory failure and death in patients with severe COVID-19, but there are more limited data for the use of tofacitinib in COVID-19 (118). However, studies on ruxolitinib, another JAK inhibitor, could not confirm a significant reduction in mortality when using it to treat severe COVID-19 (119, 120).
Combination of Immunomodulatory Drugs
The current guidelines recommend adding a second immunomodulatory drug, baricitinib or tocilizumab, for patients with severe COVID-19 with rapidly increasing oxygen needs and systemic inflammation. In the case of the unavailability of tocilizumab, sarilumab could be used as an alternative. After the demonstration of incremental survival benefit of the triple combination of dexamethasone, IL-6 receptor blockers and baricitinib in 2659 patients in the RECOVERY Baricitinib Trial the WHO recommended that these three drugs could be combined for the treatment of patients with severe COVID-19 (14, 46).
Others
Dornase alfa (inhaled DNase), apremilast (PDE4 inhibitor), celecoxib/famotidine (COX2/histamine blockade), cenicriviroc (CCR2/5 antagonist), IC14 (anti-CD14) and icatibant (bradykinin antagonist) were found to be none-effective in an RCT (121). The six-month survival of REMAP-CAP study participants was evaluated to investigate the long-term effects of the anti-inflammatory drugs used during acute COVID-19 in severe cases. Among critically ill patients with COVID-19 randomized to receive either an IL-6 receptor antagonist or an antiplatelet had an improved 180-day mortality compared with patients randomized to the control, with a 99.9% and 95.0% probability and aHR=0.74 (95% CrI=0.61-0.90) and HR=0.85 (95% CI=0.71-1.03), respectively (109).
Anticoagulant Therapy
COVID-19, especially in severe clinical forms, is associated with a hypercoagulable state, the degree of which closely related to the systemic inflammatory response during the disease. Arterial and venous in situ thrombosis in large and small blood vessels is the result of mononuclear cell activation and endothelial cell inflammation. Recommendations for antithrombotic treatment and/or doses of it differed according to the severity of COVID-19 (122-129).
Outpatient with Mild COVID-19
As the rate of thrombotic events is low and studies have not shown any benefit, anticoagulant drugs are not recommended for outpatients with mild COVID-19. In a recent placebo-controlled RCT study of 1284 non-hospitalized patients with symptomatic COVID-19 and at least one thrombosis risk factor, rivaroxaban was not more beneficial for decreasing thrombotic event, hospitalization and death than placebo with an HR of 1.16 (95% CI=0.63-2.15; p=0.63) (122).
Hospitalized Patients with Severe COVID-19 Requiring Conventional Oxygene
Findings from the studies performed during the early phase of the pandemic reported that in noncritically ill patients with COVID-19, therapeutic-dose anticoagulation with heparin is related to improved survival and reduced thrombotic events (123-125). In an RCT of noncritically ill hospitalized patients with COVID-19, mortality and endotracheal intubation rates were significantly lower among patients treated with therapeutic dose enoxaparin or therapeutic-dose apixaban compared with prophylactic-dose enoxaparin with an HR of 0.70 (95% CI=0.52-0.93; p=0.01), and (HR=0.75, 95% CI=0.58-0.98; p=0.03), respectively (126). For hospitalized patients who require conventional oxygen with a D-dimer level above normal and who do not have an increased bleeding risk, a therapeutic dose of heparin (enoxaparin) is recommended (127).
For Patients Admitted to the Intensive Care Unit with Critical COVID-19
Therapeutic dose antithrombotic therapy was not shown to be beneficial for survival or hospital discharge but related to excess bleeding events for patients with critical COVID-19 (128).
For patients with critical COVID-19 admitted into ICU and requiring high-flow nasal cannula (HFNC) oxygen or noninvasive ventilation (NIV) or MV or ECMO, prophylactic dose of heparin (enoxaparin) is recommended unless contraindicated (128).
Extended Prophylaxis After Hospital Discharge
Although extended thromboprophylaxis was previously found to be related to improved outcomes for patients discharged after COVID-19 and at increased risk for venous thromboembolism, recent studies could not find such an association. Routine post-discharge thromboprophylaxis is not recommended, but rivaroxaban 10 mg for 31 to 39 days could be considered for patients with risk factors for thrombosis, such as having a previous attack of venous thromboembolism (VTE) or major surgery or trauma (128, 129).
Antiplatelet Therapy
Current data do not support the use of antiplatelet agents, including aspirin or P2Y12 inhibitor, ticagrelor for either outpatients or inpatients with COVID-19. Systematic review and meta-analysis of four RCTs, including 17,541 hospitalized patients with moderate to severe COVID-19, revealed that antiplatelets had no effect on 28-day mortality and clinical improvement but probably resulted in a slight reduction in thrombotic events and an increase of major bleeding events (130). Recent RCTs also do not support using antiplatelets to treat COVID-19 (131).
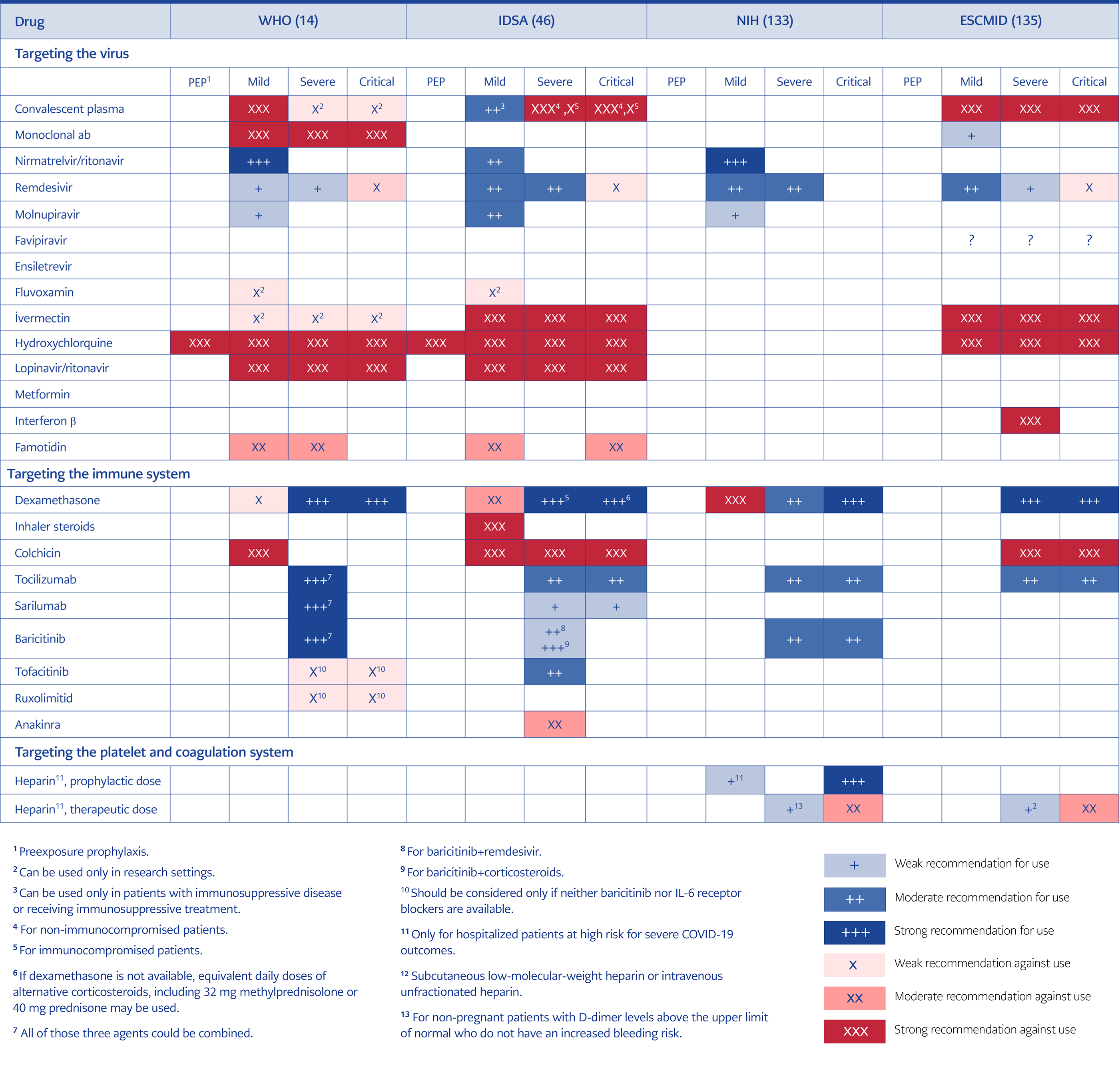
Treatment of Immunocompromised Patients Who Have Prolonged Symptoms and Positive SARS-CoV-2 Viral Test Results
The optimal management of immunocompromised patients with prolonged symptoms and SARS-CoV-2 NAAT or antigen test positivity despite the previous course of antivirals effective against SARS-CoV-2 is unknown. Additional anti-viral treatments, prolongation of duration of antiviral treatment, and high-titer CP from a donor who had recently COVID-19 with the currently circulating variant could be the options, but none of them have enough evidence at the moment (132, 133). The summary of current recommendations of the WHO (14), the US National Institute of Health (NIH) (133), IDSA (46) and the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) (134) for the treatment of COVID-19 were given in Table 2.