Introduction
Candida species are major sources of morbidity and mortality in healthcare settings. These infections are predominantly connected with medical procedures. The five most prevalent pathogens- Candida albicans, Candida glabrata, Candida tropicalis, Candida parapsilosis, and Candida krusei – are responsible for more than 90% of invasive diseases (1, 2).
Candidemia is the most common form of invasive candidiasis, and it is associated with end-organ involvement, prolonged hospitalization, increased mortality, and higher healthcare costs (3). Candida bloodstream infections can lead to metastatic heart and ocular infections. Endocarditis caused by Candida species is a serious complication with a mortality rate of about 30% (4).
This study aimed to define the incidence, demographics, distribution, clinical characteristics, and mortality of candidemia episodes and compare the current data with the previous results of our center.
Materials and Methods
In this single-center retrospective observational study, we enrolled 250 patients diagnosed with candidemia in a university hospital between January 2015 and December 2020. We obtained patients’ demographic (age, gender, comorbidities, presence of invasive device), clinical (department, duration of hospital stay, invasive devices, intensive care unit [ICU] duration, and mortality), laboratory (Candida identification and antifungal susceptibility results, kidney, and liver function tests) and therapeutic data (steroid usage, previous antibiotic usage, total parenteral nutrition, chemotherapy and other immunosuppressive treatments, antifungal agent, duration of treatment) from electronic medical records. An ophthalmologic examination and screening with echocardiography were carried out within the first week after the diagnosis of candidemia; ocular and cardiac involvements were recorded. We included only patients over 18 years. In addition, following the literature, we included only one of the duplicate cultures growing the same organism with the same susceptibility profile in the last 14 days to diagnose candidemia (5). The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist was used for the study design.
Blood cultures were incubated for up to five days in BD BACTECTM automated blood culture system (Becton Dickinson, Sparks, NV, USA). An antifungal susceptibility test, matrix-assisted laser desorption/ionization time-of-flight, and mass spectrometry (MALDITOF-MS) were used for Candida isolate identification.
We used the Shapiro-Wilk test to determine if quantitative variables were distributed normally and presented median (minimum-maximum) for quantitative variables and n (%) for categorical variables. Independent categorical data were analyzed using Pearson’s chi-square or the Fisher-Freeman-Halton exact tests. When the Fisher-Freeman-Halton exact test was used, the Mann-Whitney U test was performed to compare the vaccination groups. The statistical analyses were performed using the Statistical Package for Social Sciences (SPSS) for windows 25.0 (IBM Corp., Armonk, NY, USA), and the type I error probability was set as 0.05.
The Başkent University Institutional Review Board approved the study on January 11, 2017, with the decision number KA16/316.
Results
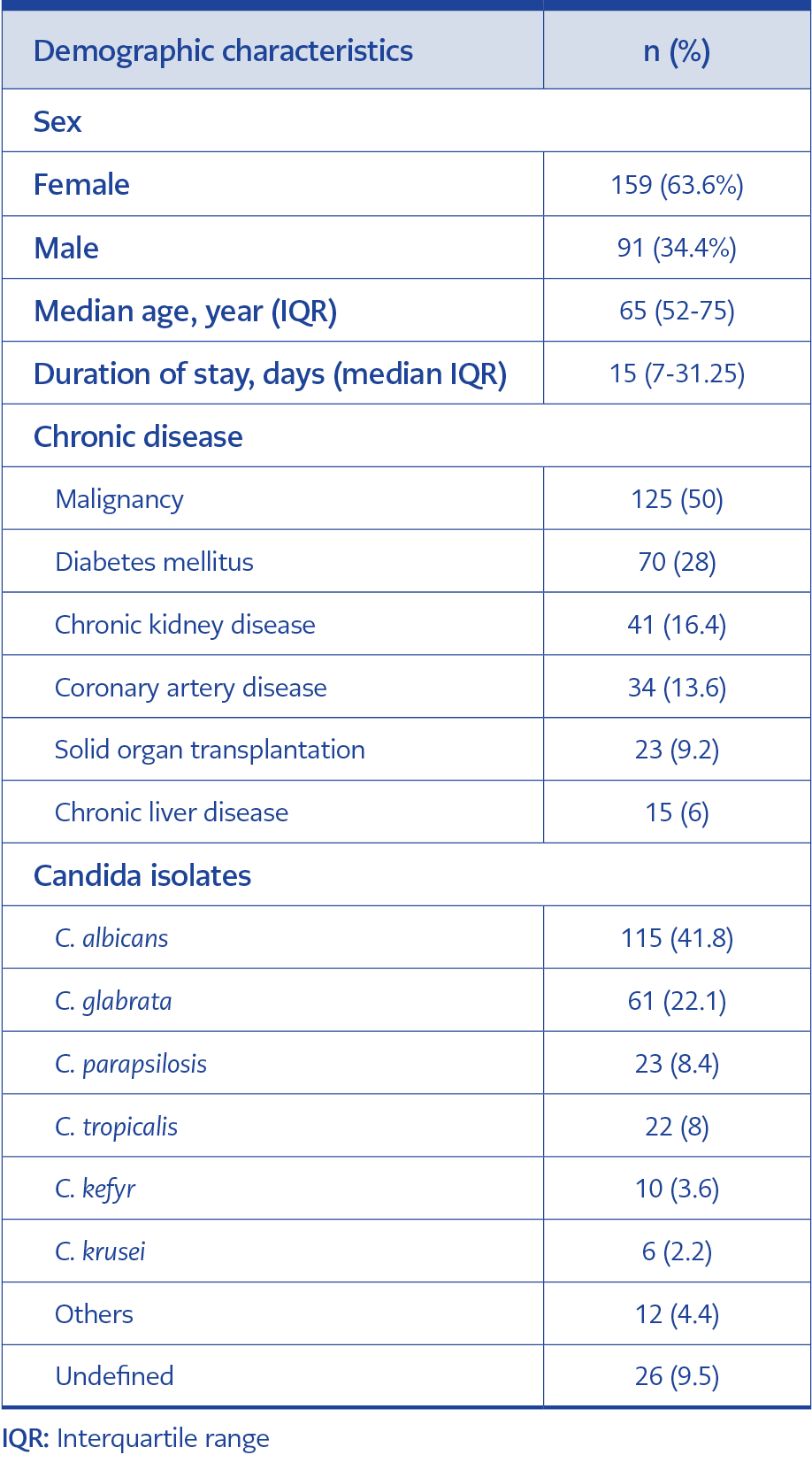
This study included 275 candidemia episodes from 250 patients over 18 years. Ninety-six thousand nine hundred one people were admitted to our center throughout the research period, with 483,560 patient days. The incidence of candidemia was 2.8% per 1000 admissions and 5.68% per 10,000 inpatient days. The median age was 65 (interquartile range [IQR]=52-75) years, and 159 (63.6%) of the patients were female. The median duration of hospital stay was 15 (IQR=7-31.25) days. The most frequent comorbidity was malignancies, involving 125 cases (50%). The demographic characteristics of the patients are shown in Table 1.
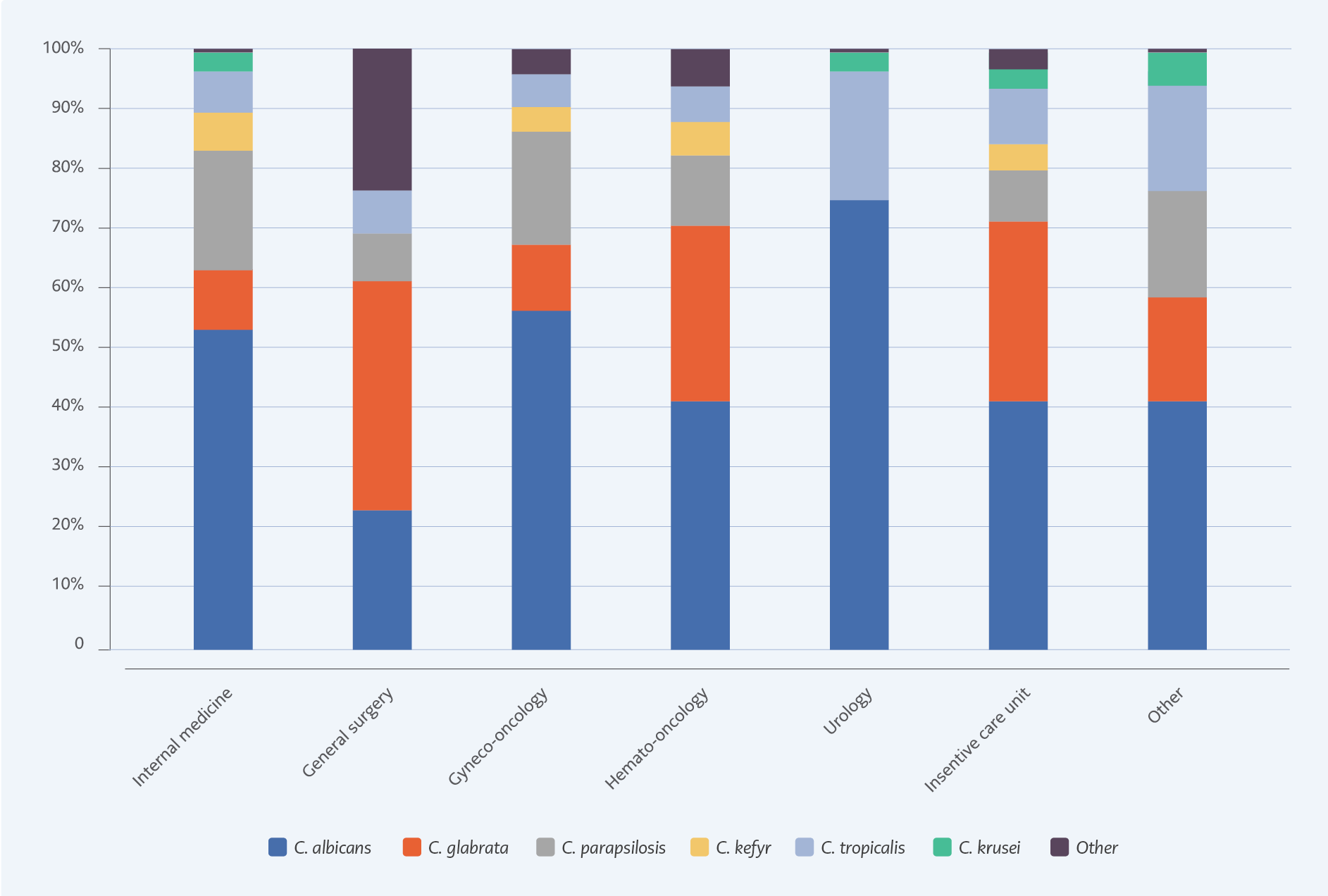
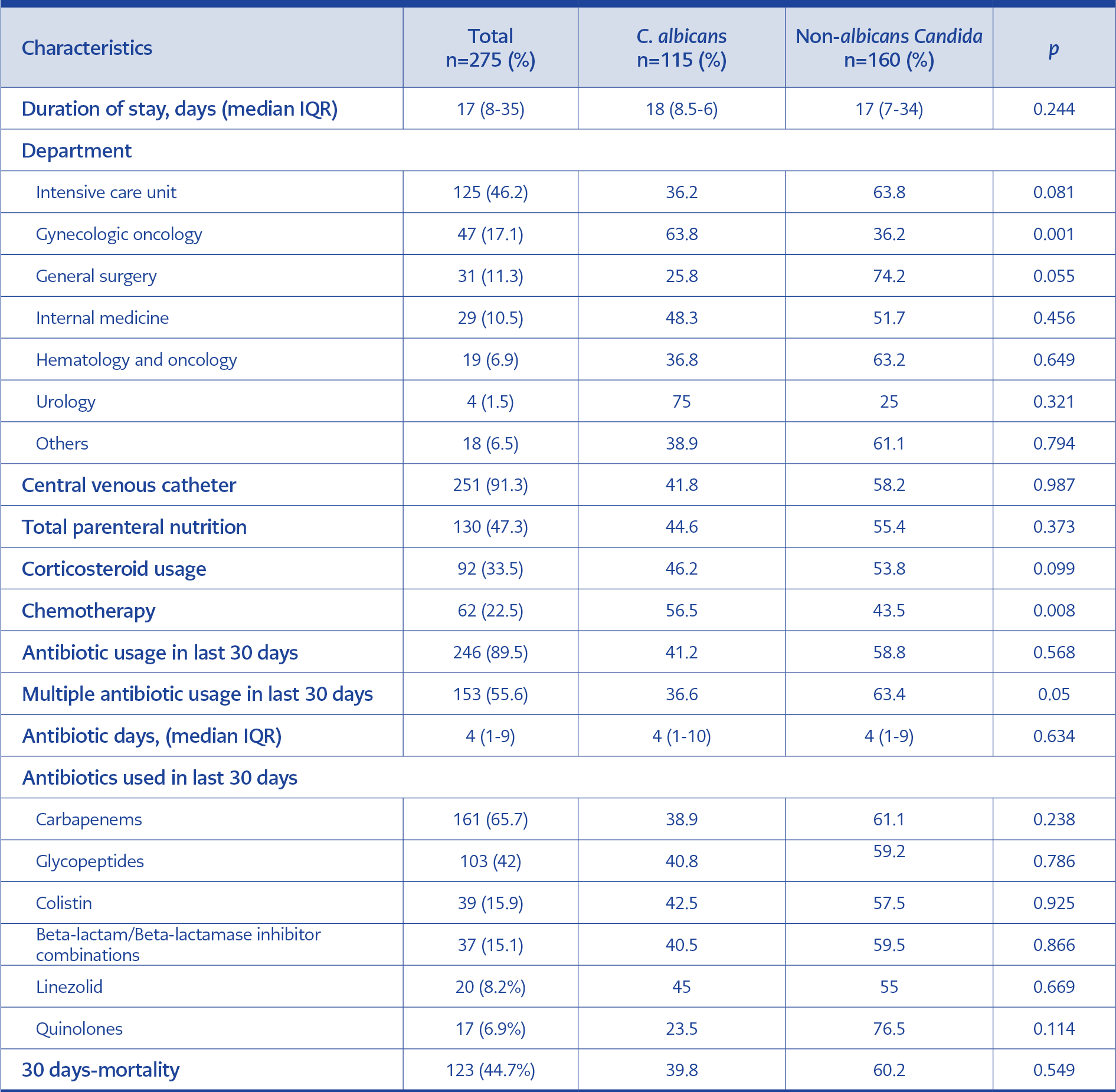
The most common Candida isolate was C. albicans (n=115, 41.8%); however, non-albicans Candida species were more than half (n=160, 58.2%). The second most common subtype was C. glabrata (n=61, 22.2%), particularly in surgical patients, patients with malignancy, and critically ill patients. Distribution of Candida isolates and distribution according to hospital ward are shown in Figures 1.
Candidemia episodes were observed most frequently in the ICU. For the episodes, the median duration of ICU stay was 17 (8-15) days. Central catheter usage was 91.3% (n=251), and in 47.3% of the episodes, patients were under total parenteral nutrition (TPN). Patients had received antibiotics within the previous 30 days in 89.5% of the episodes, and multiple antibiotic usage was 55.6%. The median duration of antibiotic treatment was four (1-9) days. The most frequently utilized antibiotic groups were carbapenems (n=161, 65.7%) and glycopeptides (n=103, 42.0%) in the last 30 days.
The catheter was removed in only 52% of patients with a central venous catheter (CVC), with a median duration of 4 (IQR=2-6) days. Follow-up blood cultures were tested in 227 (82.5%) patients, and the median time to culture negativity was 3 (IQR=2-6) days. Nearly one-quarter (n=52) of the individuals who had growth in their blood cultures from a port or central catheter also had growth in their peripheral blood cultures. There was no statistically significant difference between C. albicans and non-albicans according to neither CVC presence nor TPN (p=0952 and 0.261, respectively).
Within six months, 24 individuals experienced two episodes; the median time between episodes was 32 (IQR=20-54.75) days. The same pathogen as in the first episode was isolated from the blood in 22 (91.7%) of the second attacks.
The 93.3% (257) patients had infectious diseases consultations. The ophthalmoscopic examination was done in 145 episodes (52.7%), endophthalmitis was detected in 16 (11.0%) cases, and two-thirds of them were caused by C. albicans. Half of the patients with endophtalmic involvement died in 30 days, and one had decreased vision. In seven patients, visual improvement was seen with systemic antifungal treatment. Echocardiography was performed in 139 (50.5%) episodes; one case had an endocarditis diagnosis.
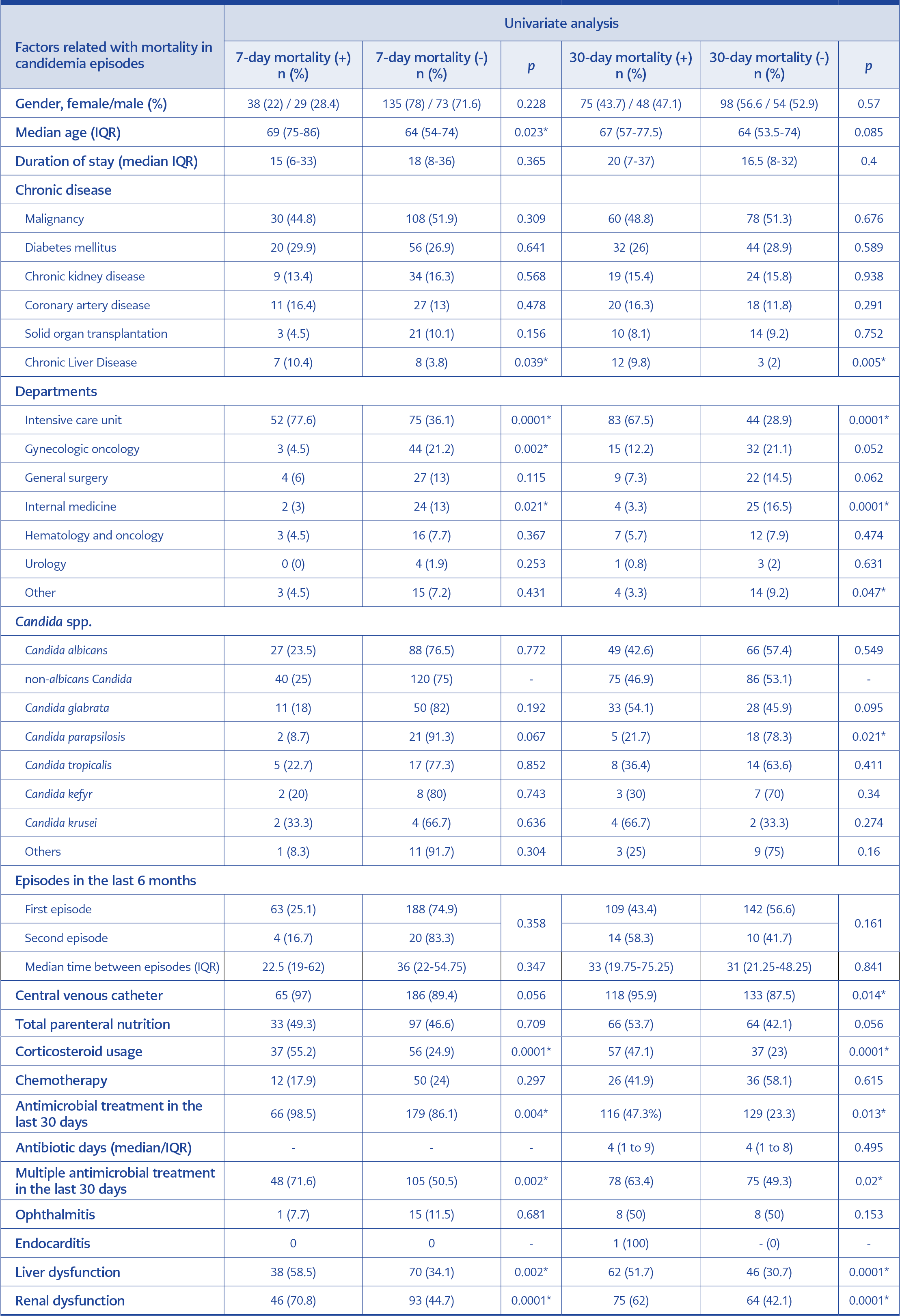
The 7-day mortality for all candidemia episodes was 24.4% (n=67), and the 30-day mortality was 44.7% (n=123). One-third (14.9%; n=41) of the deaths occurred in the first three days. The mortality rates of candidemia caused by albicans and non-albicans Candida species were not significantly different (p=0.772 and p=0.549, respectively). However, when examined independently, 30-day mortality rates in candidemia caused by C. glabrata and C. krusei were shown to be high (54.1% and 66.7%, respectively), albeit not statistically significant (p=0.095 and 0.274, respectively). In candidemia caused by C. parapsilosis, the 30-day mortality rate was significantly lower (21.7%; p=0.021).
The factors related to both 7-day and 30-day mortality were ICU requirement (p=0.0001 and p=0.0001), chronic liver disease (p=0.039 and p=0.005), corticosteroid usage (p=0.0001 and p=0.0001), previous antibiotic usage (p=0.004 and 0.013), multiple antibiotic usage (p=0.002 and 0.020), having liver (p=0.002 and p=0.001) or renal dysfunction (p=0.0001 and p=0.001). Therefore, advanced 7-day mortality was higher in older age (p=0.023), and 30-day mortality was higher in CVC-related candidemia (p=0.014). In addition, patients who received total parenteral nutrition had higher 30-day mortality (53.7% vs. 42.1%), and the mortality rate of second episodes was higher (58.3% vs. 43.4%); however, differences were not statistically significant (p=0.056 and p=0.161, respectively). Factors related to 7-day and 30-day mortality are shown in Table 3.
Discussion
The epidemiology of candidemia varies from country to country, from center to center, and even within the same center over time (6-9). We found that the incidence of candidemia episodes was 2.8 per 1000 admissions and 5.68 per 10,000 inpatient days in our center between January 2015 and December 2020. These rates were higher than our center’s 2007-2014 results, which were 1.23 episodes per 1000 admissions and 3.29 per 10,000 inpatient days (6). Similar increases have been reported in Turkey and other countries throughout the years.
For instance, studies reported that the incidence of candidemia episodes increased from 0.21 to 0.51 in Australia (10, 11), from 0.2-0.38 to 0.83 in European countries (6, 12), from 0.74 to 1.2-1.3 in Brazil (13, 14), and only in three years from 0.47 to 0.69 in Croatia (15). Although there has been a global rise throughout the years, our rates are still higher than the whole incidence (6, 12). For Turkey, different studies have reported an incidence between 0.2 and 1.8 per 1000 admissions (16-18). In Koehler et al.’s hospital-based meta-analysis in Europe, the estimated overall pooled incidence rate (IR) of candidemia was reported to be 0.83 per 1000 admissions per year, varied from 0.17 in Finland to 2.19 in Portugal. We assumed that the high incidence in our center was due to the high number of immunosuppressive patients, such as cancer patients, and the high frequency and duration of intensive care hospitalization of these patients. Unfortunately, antibiotic consumption rates were still higher than in other countries, as previously reported by our center (19).
The shift in the causative species of Candida was the second significant finding. The most often isolated strain remained C. albicans, although this frequency dropped from 54.6% to 41.8%. That change was in line with recent studies (8). C. glabrata (22.2%), C. parapsilosis (8.4%), and C. tropicalis (8%) were the top three non-albicans Candida strains in our study. These rates are similar to Korean studies and North American data from the SENTRY surveillance study (20, 21) but different from other Turkey data (22). Shifting from C. albicans to non-albicans Candida is associated with antifungal resistance, treatment failure, recurrent episodes of candidemia, and increased mortality globally (8, 21, 23). C. glabrata, more frequent in critically ill patients, has been associated in multiple studies with higher fatality rates and antifungal resistance. In line with other studies, we observed C. glabrata candidemia most commonly in ICU, with significantly higher levels of azole resistance. Fatality rates exceeded 50%, but this difference was not statistically significant.
Many studies described CVC presence, TPN, long hospitalization period and broad-spectrum antibiotic usage as the main risk factors (3, 24). In our study, CVC utilization and TPN rates were both high (91.3% and 47.3%, respectively), and the median length of ICU stay was 17 (8 to 15) days. Almost 90% of the patients had received wide-range antimicrobial therapy in the last 30 days, and more than 50% had received multiple antibiotics. Catheter removal was achieved only in 52% of cases. All of these factors might have caused high candidemia rates in our center.
In this study, although infectious diseases consultation was made in 90% of the patients with candidemia, ophthalmoscopic examination, and echocardiographic evaluation could be performed in only half of the patients. Recent studies have demonstrated a favorable impact on the course of candidemia and mortality using bundles containing standard approaches for diagnosis, follow-up, and treatments when performed under the supervision of an infectious disease specialist. Infectious disease consultation may provide updated information about the local epidemiology and resistance patterns, appropriate antifungal management, standard ophthalmoscopic and echocardiographic evaluations, early source control (such as removal of catheters), and regular patient follow-up (25, 26). Ophthalmoscopic and echocardiographic evaluations are important components of these bundles because, according to numerous studies, ocular and cardiac involvement of Candida infection is strongly linked to mortality (25, 27, 28). In our study, half of the patients with retinitis/ endophthalmitis died. However, there was no correlation between retinitis/ endophthalmitis and mortality. In one study, because endophtalmic involvements are rare and usually associated with a good outcome, it was stated that routinely performed ocular assessment might not be necessary for every patient with candidemia (29). However, in many studies, the benefits of early endophthalmitis diagnosis are related to better outcomes (25, 30). Like endophthalmitis, there was no significant difference between endocarditis and its absence. It might be due to the limited numbers. Another factor is that practically all patients who had an echocardiographic evaluation had only one examination, which increases the risk of missing the diagnosis because there is no control evaluation.
Our study has some limitations. The main limitations are the lack of antifungal susceptibility results and the small number of participants. In addition, the impact of empirical antifungal therapy on outcomes could not be evaluated. Larger cohorts are needed to evaluate the effects of a multidisciplinary approach and bundle implementations on the development of candidemia and related mortality.
In conclusion, candidemia bundle applications would be effective strategies for providing an effective antifungal stewardship program because of the life-threatening complications such as endocarditis, high mortality rates, and increased healthcare costs. Furthermore, our study shows that even consultation with infectious disease specialists seems insufficient to reduce the incidence of candidemia and mortality rates. Therefore, comprehensive candidemia bundles may improve patients’ outcomes.