Introduction
Since its emergence in December 2019, 530,266,292 confirmed coronavirus disease 2019 (COVID-19) cases, including 6,299,364 deaths were reported as of June 7, 2022 (1). It’s widely accepted that in older age, comorbidities (hypertension, diabetes mellitus, obesity, chronic respiratory disease, chronic kidney disease, cardiovascular disease, malignancies) and immunosuppression significantly increase the risk of severe COVID-19 (2, 3).
Worldwide, 37.7 million people are infected with the human immunodeficiency virus (HIV) (4). Since early in the COVID-19 pandemic, there has been a growing interest in exploring the impact of COVID-19 among people living with HIV (PLWH). Although initial studies assessing the severity of COVID-19 among PLWH do not suggest poorer outcomes, recent findings showed a higher risk of morbidity and mortality, particularly in those with lower CD4 cell counts, unsuppressed HIV viremia and comorbidities mentioned above (5, 6). Still, it is debated whether PLWH have an increased risk of severe COVID-19, as the data come from cohort studies performed with unmatched populations. Despite the existing debate, World Health Organization (WHO) Global Clinical Platform for COVID-19 announced HIV infection as an independent risk factor for severe COVID-19 (7). The susceptibility of PLWH to SARS-CoV-2 infection has also been debated. The incidence of COVID-19 among PLWH ranges between 0.3-5.7% (5, 8). Current data revealed that SARS-CoV-2 infection rates among PLWH are not higher than their HIV-negative counterparts (8, 9). It was speculated that PLWH have a lower risk of SARS-CoV-2 infection rates in terms of preventive effects of antiretroviral therapy (ART) against COVID-19 (8). In two cohort studies from Spain and South Africa, tenofovir disoproxil fumarate use was linked with lower SARS-CoV-2 infection rates (8,10). In a letter by Joob et al., where the authors shared their observation in Thailand within a high prevalence area of HIV infection, authors reported that HIV-infected patients on antiretroviral treatment (lopinavir/ritonavir) have not been infected with SARS-CoV-2 and concluded that antiretroviral treatment might be a protective factor for PLWH (11).
On the other hand, Ayerdi et al., reported higher SARS-CoV-2 seroprevalence in tenofovir disoproxil fumarate/emtricitabine users compared to the control group (12). Similarly, the Prevenir ANRS study group couldn’t find any evidence of a preventive effect of tenofovir disoproxil fumarate/emtricitabine against SARS-CoV-2 infection (13). Also, results of the studies investigating the therapeutic role of lopinavir/ritonavir and darunavir/cobicistat in SARS-CoV-2 infection were not in favor of using these antiretroviral drug combinations (14-17). Unfortunately, there is a lack of population-based studies assessing the COVID-19 susceptibility of PLWH who receive different antiretroviral molecules. Consequently, whether various antiretroviral drugs have preventive effect against SARS-CoV-2 infection or PLWH under ART have a milder course of COVID-19 still remains unanswered.
Controversial results of the studies mentioned above led us to analyze the widely used antiretroviral molecules, in particular; abacavir, cobicistat, dolutegravir, elvitegravir, emtricitabine, lamivudine, raltegravir, and tenofovir, for possible interactions with SARS-CoV-2 as well as angiotensin-converting enzyme 2 (ACE2), the protein that participates in the entry of SARS-CoV-2 into human cells.
Materials and Methods
Molecular Docking
Molecular docking studies were performed in Schrödinger Maestro version 11.5 software (Schrödinger, LLC, New York, USA) in four stages: protein preparation, ligand preparation, grid determination, and docking. Firstly, the crystal structure of the SARS-CoV-2 chimeric receptor-binding domain complexed with its receptor human ACE2 (protein data bank [PDB]: 6VW1), SARS-CoV-2 spike ectodomain structure (open state) (PDB: 6VYB), structure of the SARS-CoV-2 spike glycoprotein (closed state) (PDB: 6VXX), structure of the post-fusion core of SARS-CoV-2 S2 subunit (PDB: 6VXX and PDB: 6LXT), structure of SARS-CoV-2 RNA-dependent RNA polymerase (PDB: 6M71) and structure of SARS-CoV-2 main protease (PDB: 6LU7) were downloaded from RCSB Protein Data Bank (PDB) (https://www.rcsb.org) and prepared in three steps: preprocess, review & modify and refinement by using “Protein Preparation Wizard” module (18-22). The energy of the protein structures was minimized using OPLS3 force field. In the second stage, cobicistat, lopinavir, ritonavir, emtricitabine, lamivudine, abacavir, dolutegravir, elvitegravir, raltegravir, and tenofovir were downloaded in 3D SDF format from National Library of Medicine (NIH) PubChem (https://pubchem.ncbi.nlm.nih.gov/) and prepared using OPLS3 force field and ionization which generated possible states at target pH: 7+/-2 with Epik (Schrödinger, LLC, New York, USA) by using “LigPrep”module. In the third stage, binding site determination of SARS-CoV-2 main protease (PDB: 6LU7) was run by clicking any atom of the ligand, and the grid box was created 20x20x20 Å size. The active site of other protein structures, whose protein-ligand complex structure has not been obtained yet and whose active site is not fully known, were determined using the “Binding Site Detection” module. All possible active sites were determined and adjusted to 20x20x20 Å using the “Receptor Grid Generation” module. At the last stage, molecular docking was performed in SP (standard precision) and flexible ligand options using the “Glide Ligand Docking” module. Docking, Glide and Glide emodel scores were calculated and evaluated. Extra precisions (XP) docking was performed on antiretroviral drug molecules with high SP docking scores. Molecular mechanics with generalized born surface area (MM-GBSA) values were calculated using the ‘Prime’ module to improve the scores of molecules with high XP docking results. Finally, possible 2D and 3D interactions between ligand and protein were identified.
Results
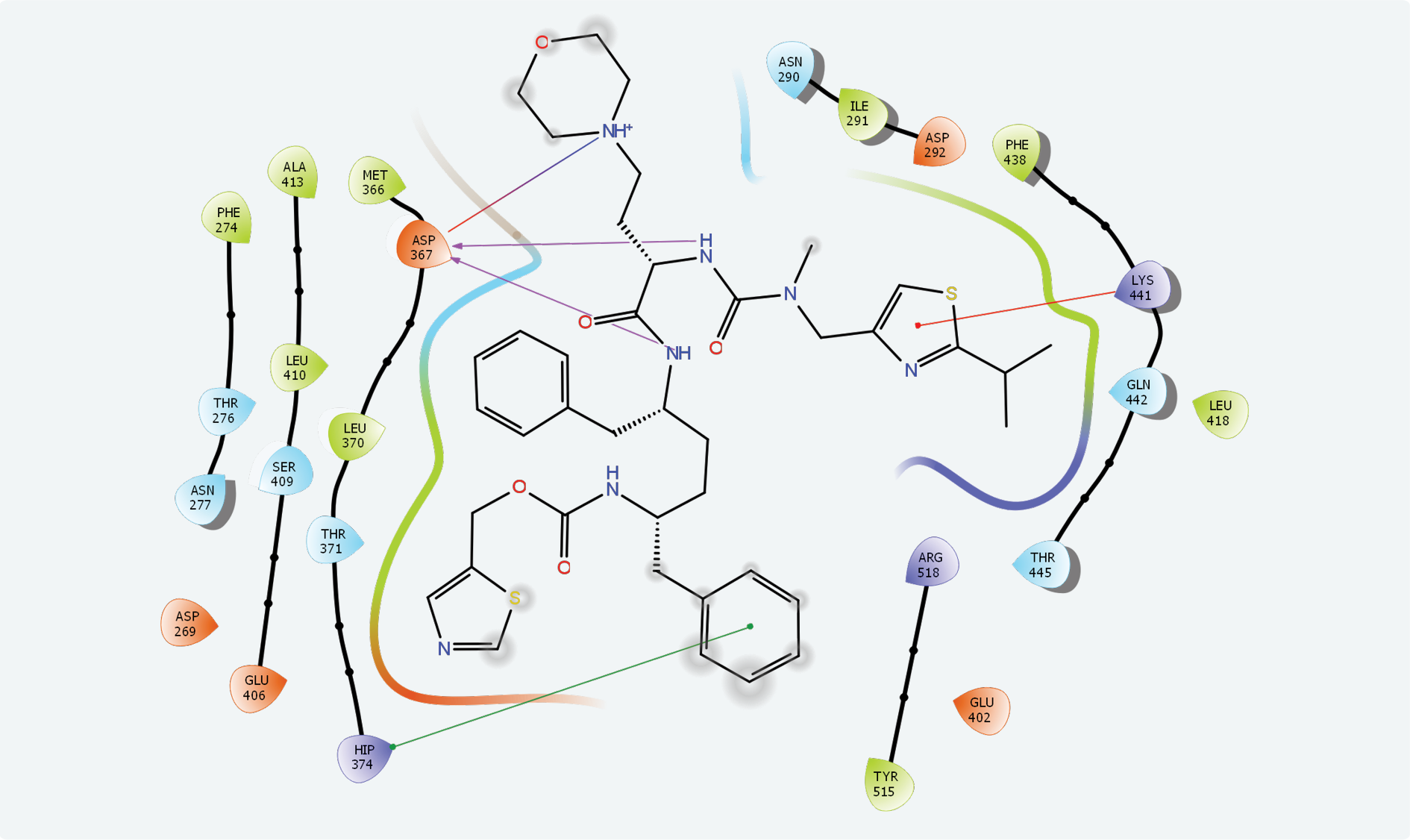
Two possible active sites have been identified on “A” chain of ACE2 by the “Binding Site Detection” module. XP docking for site 2 (x: 99.52, y: 10.48, z: 162.85) revealed the strongest interaction with cobicistat with a Glide score of -6.988 and Glide emodel score of -98.644. Ritonavir showed the second highest interaction with -6.210 and -98.377 Glide and Glide emodel scores, respectively. Compared to these scores, interactions of other molecules with active site 2 were quite low. Cobicistat and ACE2 active site had strong chemical interactions with hydrogen bonds, π–π stacking and hydrophobic interactions (Figure 1).
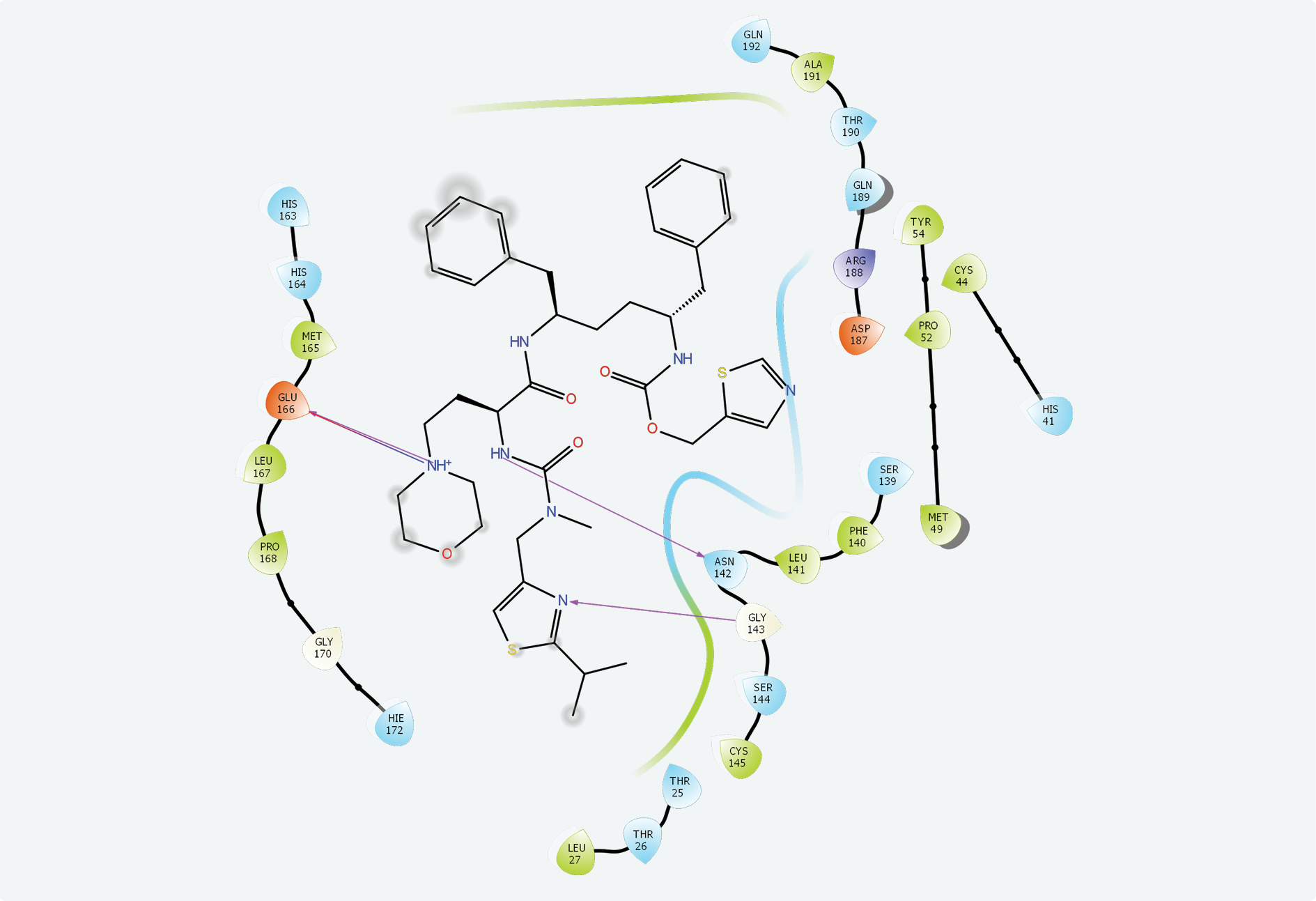
The active site of SARS-CoV-2 is known, and many protein-ligand complexes exist in PDB. Re-docking after removal of the ligand from the main protease-ligand complex 6LU7 revealed a Glide score of -8.110 and a Glide emodel score of -99.463. Cobicistat had the highest interaction with the main protease active site with a -7.763 Glide score, a -124.242 Glide emodel score and a MM-GBSA value of -105.13. Ritonavir (Glide score: -6.858, Glide emodel score: -89.094, MM-GBSA value: -100.77) and lopinavir (Glide score: -6.867, Glide emodel score: -86.508, MM-GBSA value: -71.35) also interacted with this site while none of the other compounds interacted with the active site of SARS-CoV-2. Chemical interactions between cobicistat and SARS-CoV-2 main protease active site were through hydrogen bonds and hydrophobic interactions (Figure 2).
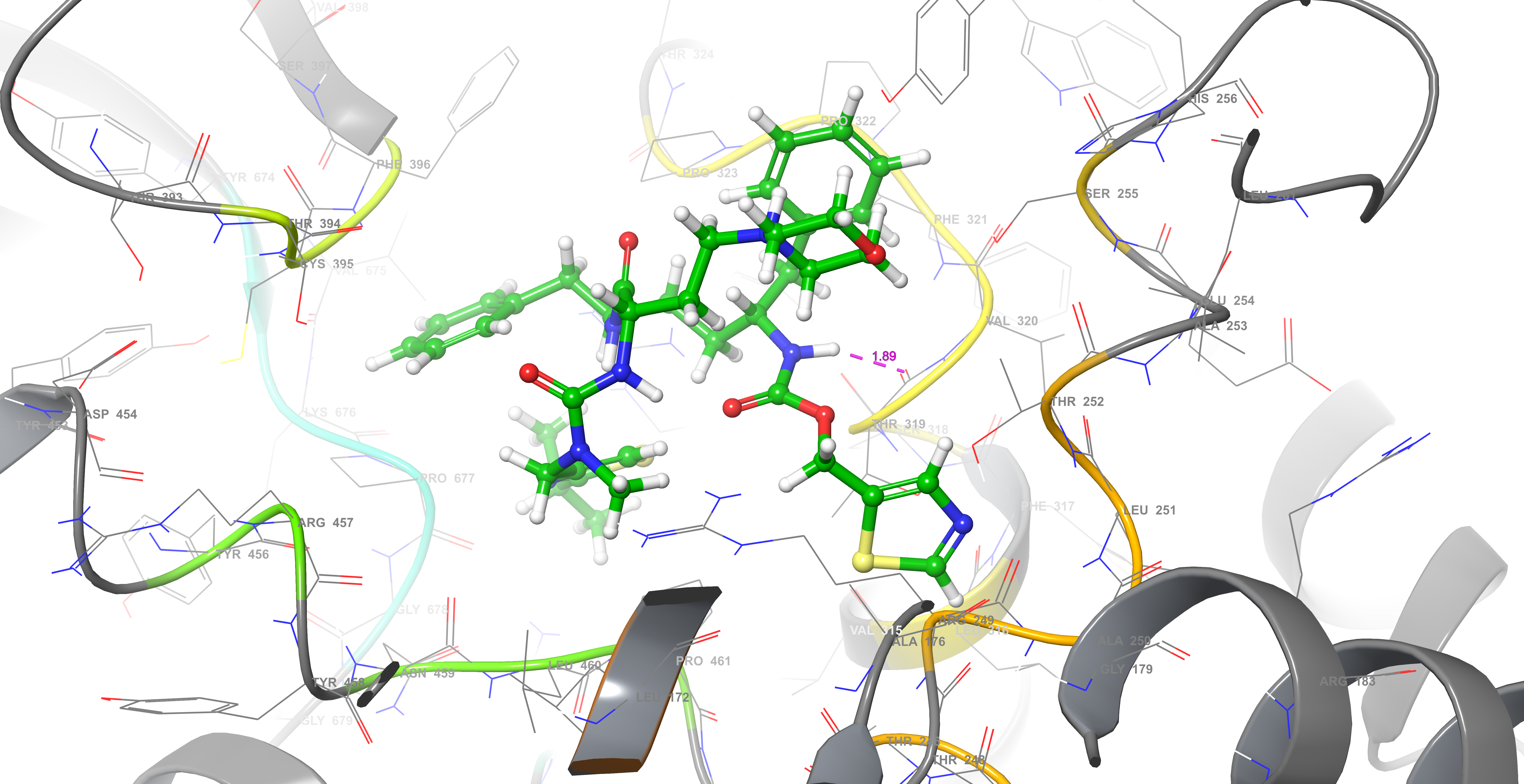
By using remdesivir, a known inhibitor of RNA polymerase (PDB code 6M71), binding site 2 was confirmed to be the active region of this enzyme. Yet, docking analyses were performed for all five binding sites on RNA polymerase. Again, SP docking showed that cobicistat has the strongest interaction with binding site 2 (XP docking score: -6.012, Glide score: – 7.639, Glide emodel score: -91.419, MM-GBSA value: -108.03). Although with higher energies, ritonavir (Glide score: -5.973, Glide emodel score: -76.862, MM-GBSA value: -78.13) also interacted with this site as while no interaction was found with other compounds. Chemical interactions between cobicistat and RNA polymerase binding site 2 were through one hydrogen bond and hydrophobic interactions (Figure 3).
In the docking procedure performed in this study, remdesivir, an RNA polymerase inhibitor, showed the highest interaction in RNA polymerase binding site 2, and other compounds other than cobicistat and ritonavir did not interact. In addition, obtaining the correct pose by re-docking the ligand to the main protease active site showed the reliability of our docking studies.
None of the drugs interacted with spike protein S1 (PDB: 6VW1_E chain), the post-fusion core of SARS-CoV-2 S2 subunit (PDB: 6LXT), SARS-CoV-2 spike glycoprotein (closed state, PDB: 6VXX) or SARS-CoV-2 spike ectodomain structure (open state, PDB: 6VYB). The results of molecular docking studies are provided as supplementary material (Supplementary Material -1).
Discussion
Finding new drugs via traditional methods is a time-consuming and expensive process. Therefore, molecular docking appears to be a useful way to find candidates from an existing library (23). To the best of our knowledge, the antiviral activity of cobicistat and ritonavir against SARS-CoV-2 has not been explored in the literature. Instead, both molecules act as pharmacokinetic enhancers by increasing the systemic exposure of co-administered antiviral agents. The present study indicated that compared to other molecules analyzed, cobicistat had the strongest interaction with SARS-CoV-2 RNA polymerase and main protease, which may suggest a rather unexpected inhibitory/antiviral effect. Ritonavir, a chemically similar antiviral agent, also interacted with these proteins with higher energy. In line with our findings, in several molecular screening studies, cobicistat and ritonavir have been proposed to be SARS-CoV-2 main protease inhibitors (24-26).
Although observational studies have indicated a potential benefit of lopinavir/ritonavir in the treatment of COVID-19 patients, RECOVERY, SOLIDARITY, and CORIST trials failed to demonstrate the clinical benefit of lopinavir/ritonavir treatment beyond standard care (14-16). Similarly, the darunavir/cobicistat combination was not found to be useful in the treatment of SARS-CoV-2 infected patients. Indeed, the authors mentioned that darunavir/cobicistat therapy was associated with an increased risk of death in two observational cohort studies, probably due to drug-drug interactions (16, 17). However, there is still a lack of randomized controlled trials assessing the effect of darunavir/cobicistat treatment among COVID-19 patients.
Pharmacologically, ACE2 is accepted as the receptor for SARS-CoV-2. Therefore, a compound that blocks ACE2 is expected to prevent SARS-CoV-2 entry into human cells. Our unexpected finding that cobicistat and ritonavir interact with ACE2 with low energy indicates that these molecules may be preventing the entry of SARS-CoV-2 by occupying its receptor. Prashantha et al., showed that both drugs have good affinity toward spike protein (27). Pfizer’s Paxlovid study, a co-packaged combination of nirmatrelvir and ritonavir, has been found to be 89% effective in patients at risk of serious COVID-19 (28). Although the scientific reports are awaited, one of the other main goals of the Paxlovid study is to assess the preventive effect of this combination on the household contacts of SARS-CoV-2 infected patients. According to our study results, ritonavir may contribute to the preventive effect of nirmatrelvir. In literature search, we could not find any clinical studies investigating cobicistat’s preventive effect against SARS-CoV-2 infection.
Our results suggest that both cobicistat and ritonavir may have preventive and therapeutic potential in SARS-CoV-2 infection. However, albeit indirect, the easiest way to determine whether this effect exists or not appears to be a long-term study in PLWH under antiretroviral regimens including these molecules.