Introduction
Despite the decrease in incidence after having widespread vaccination programs, the COVID-19 pandemic continues to cause high morbidity and mortality in the world in its second year (1). Especially elderly people (over 65 years old), people with underlying health conditions, and healthcare workers (HCWs) are still at high risk for COVID-19 disease (2). Since there is no specific treatment against SARS-CoV-2 infection, the most effective way to prevent infection is to gain immunity with an effective vaccine.
CoronaVac (Sinovac Life Sciences, Beijing, China), an inactive vaccine, is the first vaccine administered in our country. In a phase 2 study, the seroconversion rate at day 28 after CoronaVac was reported 90.7% in the 1.5 µg group, 98% in the 3-µg group, and 99% in the 6-µg group (3). We aimed to describe the SARS-CoV-2 IgG response in blood samples taken 28 days after the first dose and 28 days after the second dose from the individuals aged between 60 and 90 years who applied to our vaccine center for the administration of SARS-CoV-2 inactivated vaccine.
Materials And Methods
The 156 volunteers aged 60-90 years, who received the first dose of CoronaVac (Sinovac Life Sciences, Beijing, China) vaccine between January 14, 2021, and February 22, 2021, and the second dose of CoronaVac vaccine between February 11, 2021, and March 22, 2021, were included. At various stages, 30 volunteers left the study voluntarily. It was planned to collect peripheral blood samples from the participants 28 days after the first dose (i.e., before the second dose) and 28 days after the second dose to determine the SARS-CoV-2 IgG levels. Demographic information of all participants (age, gender, history of COVID-19, presence of comorbidities, etc.) was recorded in the follow-up form. A chemiluminescent microparticle immunoassay (CMIA)-based test (ARCHITECT IgG II Quant test, Abbott, USA) that quantitatively detects IgG antibodies specific to the RBD (receptor binding domain)/S1 region was used in the analysis of blood samples. The results obtained from all sera included in the study were evaluated as arbitrary unit/mL (AU/mL). The concentrations obtained as AU/mL were converted into the World Health Organization’s (WHO) international standard for anti-SARS-CoV-2 immunoglobulin “binding antibody unit (BAU/mL)” by multiplying with the correlation coefficient of 0.142 (4). According to this, concentrations of 50 AU/mL or 7.1 BAU/mL and above were considered positive. In addition, a concentration of 1050 AU/mL, which was reported to be 100% compatible with the plaque reduction neutralization test (PRNT), was associated with a 1:80 dilution of PRNT (5).
Statistical Analysis
The Statistical Package for Social Sciences (SPSS) version 21.0 (IBM Corp., USA) was used for analysis. Qualitative data were presented as numbers and percentages, and quantitative data as mean and standard deviation. Chi-square test and Fisher’s exact test were used in the evaluation of qualitative data, Student’s test, Mann Whitney U test, and Kruskal Wallis test were used in the comparison of quantitative data, Spearman analysis was used in correlation analysis. The p<0.05 was considered as statistical significance.
Results
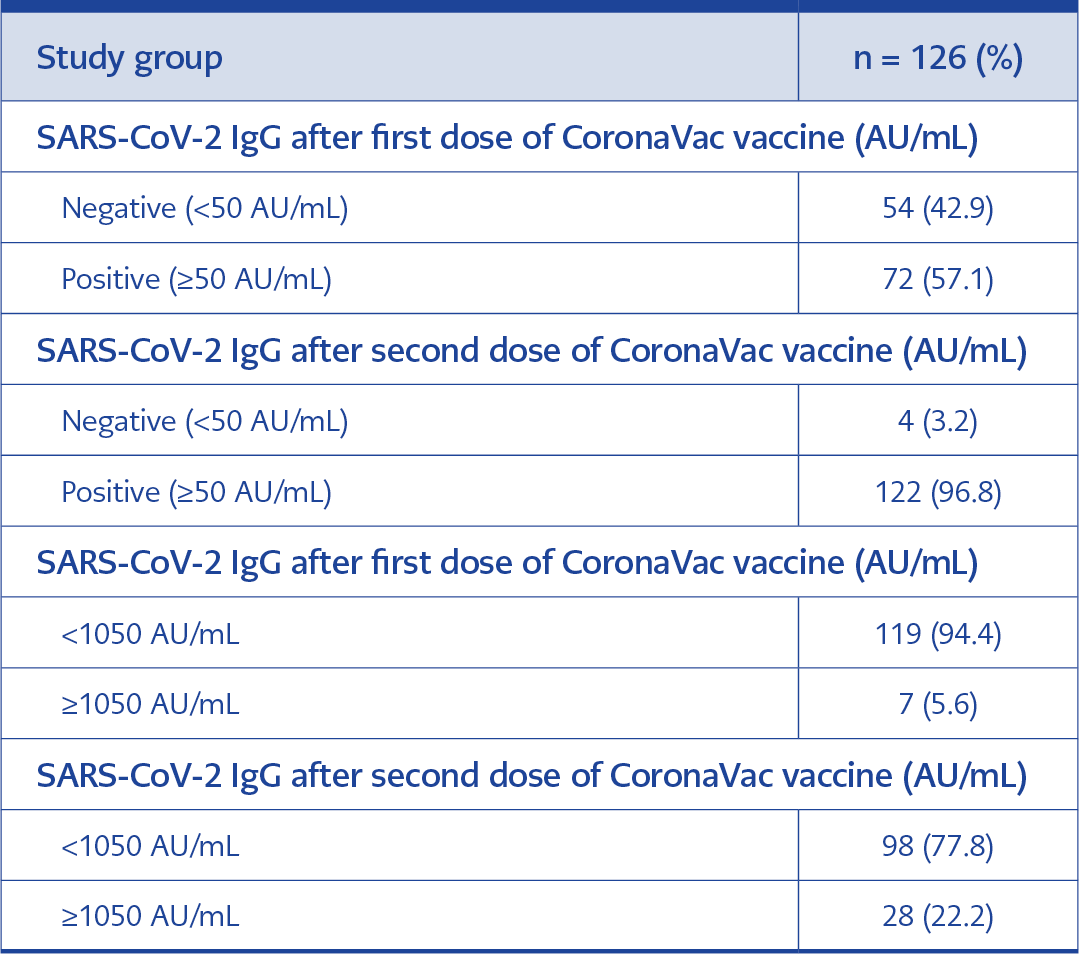
Table 1. Evaluation of the antibody results of the participants after the first and second dose vaccines as percentages.
Of the 126 participants included in the study, 66 (52.4%) were female, 60 (47.6%) were male, mean age was 69.43±5.4 (minimum 60, maximum 90 years). While no comorbidity was found in 45 (35.7%) of the participants, there were at least one comorbidity in 81 (64.3%) participants. None of the participants had a prior history of COVID-19. The antibody positivity rate was 42.8% 28 days after the first dose of vaccine, and 96.8% after 28 days after the second dose of vaccine. Four participants did not develop an antibody response (Table 1). The mean age of these patients is 72.5, and two of them had hypertension.
After two doses of vaccination, 22.2% of the participants had antibodies above 1050 AU/mL titer, which is equivalent to 1:80 dilutions in the plaque reduction neutralization test. Antibody response was not statistically significant between genders. While the median antibody titer of the participants was 43 AU/mL (Interquartile range [IQR]%:17.2-101.6) after the first dose of the vaccine, a statistically significant increase was observed after the second dose, with 561.3 AU/mL (IQR%:270.8-979.2) (p<0.001). When the antibody titers after the first and second dose were evaluated in terms of the presence of comorbidity, no significant difference was found.
Discussion
It was determined that 96.8% of the elderly individuals participating in our study developed Anti-SARS-CoV-2 IgG positivity with two doses of inactivated vaccine, but neutralizing antibody titer equivalent to 1/80 PRNT level was detected only in 22.2%.
When Karameşe et al. (6) examined the antibody titers with QuantiVac anti-SARS-CoV-2 enzyme-linked immunosorbent assay (ELISA) (IgG) (Euroimmun AG, Lubeck, Germany) in their study with 235 people aged 65 and over who were administered CoronaVac vaccine, they found the antibody titers to be 37.70 ± 57.08 IU/mL after the first dose, and 194.61 ± 174.88 IU/mL after the second dose. When they examined those below the 25.6 IU/mL value determined as negative by WHO in the same study; stated that they found this rate to be 57.02% after the first dose and 11.48% after the second dose. They stated that 120 of the 235 people included in the study had at least one comorbid condition and that the effect of diabetes mellitus (DM) on the antibody level was statistically significant (6). Our findings were similar to findings of the report by Karameşe et al., although they detected a statistical correlation between DM and antibody level only among comorbidities, no such correlation was found in our study. This may be due to the lower number of patients with DM in our study.
Our knowledge about vaccines is that the response would be lower among obese people, both in case of vaccination and exposure to infections (7). However, regarding SARS-CoV-2, the vaccine efficacy data of three FDA-approved SARS-CoV-2 vaccines (Pfizer-BioNTech, Moderna, and Johnson & Johnson) in obese and non-obese individuals in multicenter and large-participation studies did not report the difference (8). Malavazos et al. (7) found that abdominal obese individuals without a history of COVID-19 and vaccinated with BNT162b2 mRNA vaccines (Pfizer-BioNTech) produced a lower antibody response, however overweight individuals produced higher antibody titers, similar to our results after the first vaccination. However, no statistically significant results were obtained between non-obese individuals and obese individuals who were vaccinated with the CoronaVac.
In the study of Seyahi et al. (9), when they detected the antibody levels after the 2nd dose using the Elecsys kit in 47 people aged 65 and over, whom they included in the study as a control group, they reported that 14.9% of these individuals have a high antibody titer above 250 U/mL, a moderate antibody titer in the range of 117-250 U/mL in 19.1%, and a low antibody titer below 117 U/mL in 66% of the study population. In another study conducted among 24 HCWs over 60 years of age without a prior history of COVID-19 infection, an antibody response at the level of 38.2 AU/mL was detected in 9 (37.5%) of the participants 28 days after the CoronaVac vaccine. In the same study, 21 days after the second dose, they reported that 22 (95.7%) of them produced an antibody response at the level of 733.5 AU/mL (10). The findings of these reports were similar to the results of our study.
The lack of the pre-vaccine antibody titers was one of the limitations of our study. On the other hand, our study is important in terms of comorbidity, equal distribution between groups in terms of gender and BMI, the absence of a history of COVID-19 infection of the participants, and the antibody response levels to the vaccine in elderly people who were vaccinated with inactive SARS-CoV-2 vaccine for two doses.
The findings of our study reveal that neutralizing antibody levels are relatively low in elderly individuals. Within the framework of these findings, it is recommended that the third dose of vaccine is needed for the continuation of the protective immune response in elderly individuals, to prevent infection.